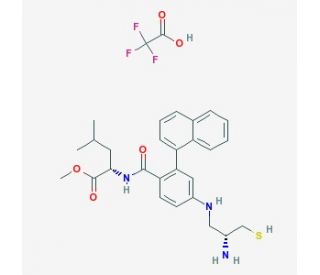
Molecular structure of GGTI 298, CAS Number: 1217457-86-7
GGTI 298 (CAS 1217457-86-7)
See product citations (2)
Alternate Names:
N-[[4-(2-(R)-Amino-3-mercaptopropyl)amino]-2-naphthylbenzoyl]leucine methyl ester trifluoroacetate salt
Application:
GGTI 298 is a CAAZ peptidomimetic GGTase I inhibitor
CAS Number:
1217457-86-7
Purity:
≥95%
Molecular Weight:
593.66
Molecular Formula:
C27H33N3O3S•CF3CO2H
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
GGTI 298 may act to be a CAAZ peptidomimetic GGTase I (geranylgeranyltransferase I) inhibitor. This compound may act to cause G0-G1 cell cycle block and apoptosis in A549 cells. GGTI 298 has demonstrated potent inhibition of GSK3 with an IC50 of 0.2 µM, exhibiting high selectivity for GSK3 over other protein kinases. It has been widely employed in various in vitro studies, including cell-based assays like cell proliferation, apoptosis, and cell migration. Moreover, GGTI298 has been utilized in biochemical assays involving enzymatic activity, protein-protein interactions, and DNA-protein interactions, showcasing its versatility and utility in research applications.
GGTI 298 (CAS 1217457-86-7) References
- The geranylgeranyltransferase I inhibitor GGTI-298 induces hypophosphorylation of retinoblastoma and partner switching of cyclin-dependent kinase inhibitors. A potential mechanism for GGTI-298 antitumor activity. | Sun, J., et al. 1999. J Biol Chem. 274: 6930-4. PMID: 10066746
- Protein geranylgeranylation is required for osteoclast formation, function, and survival: inhibition by bisphosphonates and GGTI-298. | Coxon, FP., et al. 2000. J Bone Miner Res. 15: 1467-76. PMID: 10934645
- Farnesylated RhoB prevents cell cycle arrest and actin cytoskeleton disruption caused by the geranylgeranyltransferase I inhibitor GGTI-298. | Allal, C., et al. 2002. Cell Cycle. 1: 430-7. PMID: 12548020
- Selective inhibition of cancer cell invasion by a geranylgeranyltransferase-I inhibitor. | Kusama, T., et al. 2003. Clin Exp Metastasis. 20: 561-7. PMID: 14598891
- Phosphatidylinositol-3-OH kinase/AKT and survivin pathways as critical targets for geranylgeranyltransferase I inhibitor-induced apoptosis. | Dan, HC., et al. 2004. Oncogene. 23: 706-15. PMID: 14737105
- A yeast-based genomic strategy highlights the cell protein networks altered by FTase inhibitor peptidomimetics. | Porcu, G., et al. 2010. Mol Cancer. 9: 197. PMID: 20653956
- Endothelial protective genes induced by statin are mimicked by ERK5 activation as triggered by a drug combination of FTI-277 and GGTI-298. | Chu, UB., et al. 2015. Biochim Biophys Acta. 1850: 1415-25. PMID: 25829196
- Geranylgeranyl transferase 1 inhibitor GGTI‑298 enhances the anticancer effect of gefitinib. | Liu, BS., et al. 2018. Mol Med Rep. 18: 4023-4029. PMID: 30106149
- Platelet-derived growth factor receptor tyrosine phosphorylation requires protein geranylgeranylation but not farnesylation. | McGuire, TF., et al. 1996. J Biol Chem. 271: 27402-7. PMID: 8910319
- Protein geranylgeranylation, not farnesylation, is required for the G1 to S phase transition in mouse fibroblasts. | Vogt, A., et al. 1996. Oncogene. 13: 1991-9. PMID: 8934546
- GGTI-298 induces G0-G1 block and apoptosis whereas FTI-277 causes G2-M enrichment in A549 cells. | Miquel, K., et al. 1997. Cancer Res. 57: 1846-50. PMID: 9157972
- The geranylgeranyltransferase-I inhibitor GGTI-298 arrests human tumor cells in G0/G1 and induces p21(WAF1/CIP1/SDI1) in a p53-independent manner. | Vogt, A., et al. 1997. J Biol Chem. 272: 27224-9. PMID: 9341167
- Inhibition of Ras and related G-proteins as a therapeutic strategy for blocking malignant glioma growth. | Bredel, M., et al. 1998. Neurosurgery. 43: 124-31; discussion 131-2. PMID: 9657198
Inhibitor of:
ARHGAP11B, ARHGAP15, C8orf80, Cntnap5c, CRB3, DSCR 5, Ect2, EG331493, EG432798, Enzyme, FTβ, GGPS1 (geranylgeranyl diphosphate synthase 1), GGTase, GGtase-α, GGTase-Iα, GTPBP6, Lamin A, Lamin A/C, LARG, Malcavernin, MVD, PCYOX1L, PIG-V, PIG-Z, PLEKHG1, PMVK, R-ras3, Rab 1B, Rab 2A, Rab 30, Rab 35, Rab GTPase (RAB; RAS oncogene), Rab L3, Rab40B, RABAPTIN-5, Ral GDS, RAP, RASA4, RASL11A, RCE1, REP-2, Repac, RGL4, RIN1, RSU1, TBC1D13, TBC1D2, TBC1D2B, TC 21, TERE1, TGN38B, XG FTase, and ZDHHC12.Activator of:
ACTR-IC, ARHGEF38, ARL15, ARL4D, CMV gH, FAM18A, GGtase-α, GGTase-Iα, GP73, GTPBP5, GTPBP6, H-Ras, NKHC1, PIG-Q, PRK1, R-Ras, RAM, Rap 1A, Rho GAP p190, Rho GAP p190-B, Rhotekin, and TES.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
GGTI 298, 1 mg | sc-361184 | 1 mg | $193.00 | |||
GGTI 298, 5 mg | sc-361184A | 5 mg | $838.00 |
