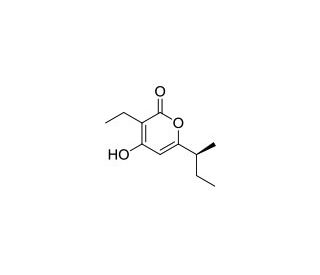
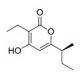
Germicidin A (CAS 151271-57-7)
QUICK LINKS
Germicidin A, a synthetic antimicrobial compound, has been specifically designed for germ eradication purposes. The active ingredient in germicidin is a quaternary ammonium compound (QAC), available in both liquid and powder forms. Extensive scientific research has extensively investigated germicidin, employing it to disinfect surfaces, water, and air in laboratory experiments. Additionally, it has been utilized to evaluate the efficacy of disinfectants against diverse microorganisms. The mode of action for germicidin involves disrupting the cell membrane of microorganisms. By binding to the cell membrane, it interferes with the microorganism′s nutrient absorption and waste expulsion mechanisms, ultimately leading to its demise. Furthermore, germicidin exhibits inhibitory effects on the growth of bacteria, fungi, and viruses, augmenting its antimicrobial properties.
Germicidin A (CAS 151271-57-7) References
- Type III polyketide synthase beta-ketoacyl-ACP starter unit and ethylmalonyl-CoA extender unit selectivity discovered by Streptomyces coelicolor genome mining. | Song, L., et al. 2006. J Am Chem Soc. 128: 14754-5. PMID: 17105255
- Physiological role of germicidins in spore germination and hyphal elongation in Streptomyces coelicolor A3(2). | Aoki, Y., et al. 2011. J Antibiot (Tokyo). 64: 607-11. PMID: 21792209
- A two-step sulfation in antibiotic biosynthesis requires a type III polyketide synthase. | Tang, X., et al. 2013. Nat Chem Biol. 9: 610-5. PMID: 23912167
- A Streptomyces coelicolor host for the heterologous expression of Type III polyketide synthase genes. | Thanapipatsiri, A., et al. 2015. Microb Cell Fact. 14: 145. PMID: 26376792
- Germicidins H-J from Streptomyces sp. CB00361. | Ma, M., et al. 2017. J Antibiot (Tokyo). 70: 200-203. PMID: 27507632
- Biomimetic iterative method for polyketide synthesis. | Akagawa, K. and Kudo, K. 2017. Chem Commun (Camb). 53: 8645-8648. PMID: 28617504
- The MtrAB two-component system controls antibiotic production in Streptomyces coelicolor A3(2). | Som, NF., et al. 2017. Microbiology (Reading). 163: 1415-1419. PMID: 28884676
- α-Pyrone Polyketides from Streptomyces ambofaciens BI0048, an Endophytic Actinobacterial Strain Isolated from the Red Alga Laurencia glandulifera. | Rab, E., et al. 2017. Mar Drugs. 15: PMID: 29240664
- Secondary Metabolites Produced during the Germination of Streptomyces coelicolor. | Čihák, M., et al. 2017. Front Microbiol. 8: 2495. PMID: 29326665
- Biosynthesis of Polyketides in Streptomyces. | Risdian, C., et al. 2019. Microorganisms. 7: PMID: 31064143
- [A new polyketide from marine-derived Streptomyces sp.MDW-06]. | Wang, C., et al. 2019. Zhongguo Zhong Yao Za Zhi. 44: 2090-2095. PMID: 31355566
- Hexokinase II Inhibitory Effect of Secondary Metabolites Derived from a Streptomyces sp. Associated with Mud Dauber Wasp. | Zhang, XM., et al. 2020. Chem Biodivers. 17: e2000140. PMID: 32515903
- 6S-Like scr3559 RNA Affects Development and Antibiotic Production in Streptomyces coelicolor. | Bobek, J., et al. 2021. Microorganisms. 9: PMID: 34683325
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Germicidin A, 1 mg | sc-362022 | 1 mg | $208.00 |
