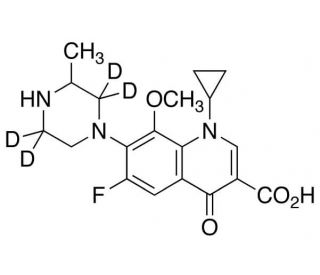
Gatifloxacin-d4
QUICK LINKS
Gatifloxacin-d4 is a deuterated analogue of gatifloxacin, where four hydrogen atoms are replaced with deuterium, a heavier isotope. This subtle modification is crucial in analytical chemistry, particularly in the study of the kinetic and dynamic behaviors of the parent molecule, gatifloxacin, under various experimental conditions. Employed chiefly in research laboratories, Gatifloxacin-d4 is utilized as an internal standard for precise quantification via high-resolution mass spectrometry. This approach enhances the accuracy of detecting and quantifying the non-deuterated form in complex biological matrices by minimizing issues related to matrix effects and ion suppression. The incorporation of deuterium increases the molecular stability and reduces the rate of degradation, which is particularly advantageous in long-term studies where the integrity of the molecule is paramount. Additionally, Gatifloxacin-d4 is instrumental in metabolic studies where tracking the biotransformation pathways of the parent molecule is required. Researchers rely on this labeled compound to distinguish between metabolites derived from the parent compound and other similar substances, thereby mapping out metabolic routes with greater specificity. This robust tool enables scientists to explore and explain the environmental fate and behavior of gatifloxacin, providing valuable insights into its lifecycle and interactions within various ecosystems.
Gatifloxacin-d4 References
- Interaction of gatifloxacin with efflux transporters: a possible mechanism for drug resistance. | Kwatra, D., et al. 2010. Int J Pharm. 395: 114-21. PMID: 20573570
- Gatifloxacin-induced histamine release and hyperglycemia in rats. | Ishiwata, Y. and Yasuhara, M. 2010. Eur J Pharmacol. 645: 192-7. PMID: 20674567
- High-resolution mapping of fluoroquinolones in TB rabbit lesions reveals specific distribution in immune cell types. | Blanc, L., et al. 2018. Elife. 7: PMID: 30427309
- Fluoroquinolone Efficacy against Tuberculosis Is Driven by Penetration into Lesions and Activity against Resident Bacterial Populations. | Sarathy, J., et al. 2019. Antimicrob Agents Chemother. 63: PMID: 30803965
- Comparison of the antibacterial activities of the quinolones Bay 12-8039, gatifloxacin (AM 1155), trovafloxacin, clinafloxacin, levofloxacin and ciprofloxacin. | Bauernfeind, A. 1997. J Antimicrob Chemother. 40: 639-51. PMID: 9421311
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Gatifloxacin-d4, 1 mg | sc-218565 | 1 mg | $360.00 |
