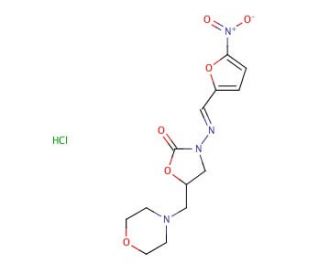
Furaltadone Hydrochloride (CAS 3759-92-0)
QUICK LINKS
Furaltadone hydrochloride, an antibacterial agent belonging to the nitrofuran class, has garnered attention in scientific research due to its unique mechanism of action and potential applications in various fields. Primarily, furaltadone exerts its antimicrobial effects by interfering with bacterial nucleic acid synthesis. Upon entering bacterial cells, it undergoes metabolic activation, forming reactive intermediates that damage microbial DNA. These intermediates lead to DNA strand breaks, inhibiting replication and transcription processes essential for bacterial survival. Consequently, furaltadone demonstrates broad-spectrum activity against Gram-positive and Gram-negative bacteria, making it a valuable tool in microbiological research. Beyond its antimicrobial properties, furaltadone hydrochloride has found applications in environmental studies, particularly in water quality assessment and monitoring. Researchers utilize furaltadone as a tracer compound to investigate contamination sources, evaluate the efficiency of water treatment processes, and assess microbial resistance in aquatic ecosystems. Furthermore, furaltadone hydrochloride serves as a model compound in pharmacokinetic and toxicological studies, facilitating the exploration of its absorption, distribution, metabolism, and excretion profiles in biological matrices. Overall, furaltadone hydrochloride emerges as a versatile chemical entity with diverse research applications spanning microbiology, environmental science, and pharmacology.
Furaltadone Hydrochloride (CAS 3759-92-0) References
- Resolution of ternary mixtures of nitrofurantoin, furaltadone and furazolidone by partial least-square analysis to the spectrophotometric signals after photo-decomposition. | Mahedero, MC., et al. 2002. J Pharm Biomed Anal. 29: 477-85. PMID: 12062648
- Metal-Organic Frameworks Meet Molecularly Imprinted Polymers: Insights and Prospects for Sensor Applications. | Lahcen, AA., et al. 2022. ACS Appl Mater Interfaces. 14: 49399-424. PMID: 36315467
- Label free detection of multiple trace antibiotics with SERS substrates and independent components analysis. | Limwichean, S., et al. 2023. Spectrochim Acta A Mol Biomol Spectrosc. 295: 122584. PMID: 36913899
- Two {CuI[P4Mo6]2}-Based Coordination Polymers Incorporating In Situ Converted Tetrapyridyl Ligands for Trace Analysis of Nitrofuran Antibiotics. | Liu, XH., et al. 2024. Inorg Chem. 63: 9058-9065. PMID: 38720438
- Effect of furaltadone on strangles in horses. | Evers, WD. 1968. J Am Vet Med Assoc. 152: 1394-8. PMID: 5689879
- Furaltadone hydrochloride in treatment of avian vibrionic hepatitis and chronic respiratory disease complex in chickens. | Eleazer, TH. and Bierer, BW. 1967. Poult Sci. 46: 819-22. PMID: 6064476
- Structural Characterization of Channel Inclusion Compounds Formed by Furaltadone Hydrochloride: Comparison to the Crystal and Molecular Structures of Furaltadone Base and Moxnidazole Hydrochloride1. | Goldberg, Israel. 1982. Journal of the American Chemical Society. 104.25: 7077-7084.
- Polarographic behaviour and determination of furaltadone in its formulations, milk and urine by differential-pulse polarography. | Díaz, T. Galeano, et al. 1993. Analytica chimica acta. 273.1-2: 351-359.
- Analysis of furazolidone and furaltadone in chicken tissues and eggs using a modified HPLC/ELCD method. | Alawi, M. A. 2000. Fresenius Environmental Bulletin. 9.7/8: 508-514.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Furaltadone Hydrochloride, 1 g | sc-211548 | 1 g | $190.00 |
