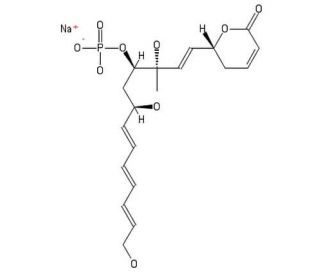
Molecular structure of Fostriecin, CAS Number: 87860-39-7
Fostriecin (CAS 87860-39-7)
See product citations (9)
Alternate Names:
Phosphotrienin; Fostriecin Sodium Salt
Application:
Fostriecin is a Topo II, PP1, and PP2A inhibitor
CAS Number:
87860-39-7
Purity:
≥95%
Molecular Weight:
452.37
Molecular Formula:
C19H26O9P•Na
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Fostriecin is an antibiotic that can be found in Streptomyces pulveraceus. This compound may act to inhibit the catalytic activity of partially purified Topo II (type II topoisomerase) in a non-competitive manner. Unlike other known inhibitors of this enzyme, fosteriecin has not been observed to cause DNA strand breaks in L1210 cells, suggesting that it does not stabilize a cleavable complex. Furthermore, in the same experiment, this agent was demonstrated to cause a block in the G2 phase of the cell cycle. Fostriecin has also displayed a strong capacity to inhibit protein phosphatase 1 (PP1) and protein phosphatase 2A (PP2A).
Fostriecin (CAS 87860-39-7) References
- Phase I and pharmacokinetic study of the topoisomerase II catalytic inhibitor fostriecin. | de Jong, RS., et al. 1999. Br J Cancer. 79: 882-7. PMID: 10070885
- Versatile enantiocontrolled synthesis of (+)-fostriecin. | Esumi, T., et al. 2002. Chem Commun (Camb). 3042-3. PMID: 12536807
- Catalyst-controlled asymmetric synthesis of fostriecin and 8-epi-fostriecin. | Maki, K., et al. 2005. J Am Chem Soc. 127: 17111-7. PMID: 16316259
- Antitumor antibiotic fostriecin covalently binds to cysteine-269 residue of protein phosphatase 2A catalytic subunit in mammalian cells. | Takeuchi, T., et al. 2009. Bioorg Med Chem. 17: 8113-22. PMID: 19857968
- Convergent synthesis of fostriecin via selective alkene couplings and regioselective asymmetric dihydroxylation. | Robles, O. and McDonald, FE. 2009. Org Lett. 11: 5498-501. PMID: 19902969
- Elucidation of the biosynthetic gene cluster and the post-PKS modification mechanism for fostriecin in Streptomyces pulveraceus. | Kong, R., et al. 2013. Chem Biol. 20: 45-54. PMID: 23352138
- Synthetic Strategies Employed for the Construction of Fostriecin and Related Natural Products. | Trost, BM., et al. 2016. Chem Rev. 116: 15035-15088. PMID: 28027648
- Effects of fostriecin on β2-adrenoceptor-driven responses in human mast cells. | Bastan, R., et al. 2017. J Immunotoxicol. 14: 60-65. PMID: 28090813
- Inhibition of type II topoisomerase by fostriecin. | Boritzki, TJ., et al. 1988. Biochem Pharmacol. 37: 4063-8. PMID: 2847752
- Total and Formal Syntheses of Fostriecin. | Dong, G., et al. 2020. Org Chem Front. 7: 3608-3615. PMID: 33184589
- Chromosome condensation induced by fostriecin does not require p34cdc2 kinase activity and histone H1 hyperphosphorylation, but is associated with enhanced histone H2A and H3 phosphorylation. | Guo, XW., et al. 1995. EMBO J. 14: 976-85. PMID: 7889943
- Antitumor drug fostriecin inhibits the mitotic entry checkpoint and protein phosphatases 1 and 2A. | Roberge, M., et al. 1994. Cancer Res. 54: 6115-21. PMID: 7954457
- Fostriecin, an antitumor antibiotic with inhibitory activity against serine/threonine protein phosphatases types 1 (PP1) and 2A (PP2A), is highly selective for PP2A. | Walsh, AH., et al. 1997. FEBS Lett. 416: 230-4. PMID: 9373158
- Renal toxicity of the anticancer drug fostriecin. | de Jong, RS., et al. 1998. Cancer Chemother Pharmacol. 42: 160-4. PMID: 9654117
- Fostriecin, an inhibitor of protein phosphatase 2A, limits myocardial infarct size even when administered after onset of ischemia. | Weinbrenner, C., et al. 1998. Circulation. 98: 899-905. PMID: 9738645
Inhibitor of:
β3Gn-T9, 4921523A10Rik, 4932429P05Rik, ALP, Aminopeptidase P1, C14orf124, C7orf47, calicin, CCDC159, CCDC25, CD92, Cdc14b Phosphatase, CMPK, CPPED1, CTDSPL, demethylated-PP2A-C, DUSP20, FASP1, FEM-2, H2-M10.2, H2-T3, H2al2y, Histone cluster 1 H2AO, Histone cluster 1 H4K, Histone cluster 2 H3B, HMBOX1, HS2ST1, HS6ST3, IPP-1, Keratin 42, Krox-8, L-Alkaline phosphatase, LCMT1, Lipocalin-11, LLGL2, LOC100039269, LOC100040299, LOC100041102, LOC100041296, LOC345630, LPPR3, MIPP, MPPE1, MPPED1, NOSTRIN, OCRL, Okadaic Acid, OS4, OST-PTP, PACE4, PAP-2c, PHACTR3, PHI-1, PHLPPL, phosphatasefamily surface anchored protein, PME-1, PP1, PP2A-β, PP2A-δ, PP2A-γ, PP2A-Aα, PP2A-Bα, PP2A-B55-β, PP2A-B55-δ, PP2A-B55-γ, PP2A-B56-β, PP2A-B56-δ, PP2A-B72/B130, PP2A-C, PP2A-Cβ, PP2C, PP2Cε, PP6, PPEF-1, PPM1H, PPP1R14D, PPP1R3, PPP1R6, PPP2R3A, PPP2R3C, PPP2R5E, Ppp4r1l, PPP4R4, PRIP, PTPLAD2, REEP2, Sds22, SH-PTP1, Sialyltransferase 7F, TMPRSS11D, Topo II, Topo IIIβ, TSPAN18, TSPY4, Type I 4-phoshatase α, Type I 4-phoshatase β, Type II 4-phoshatase, Type II 4-phoshatase β, ZDHHC9, and ZNF279.Activator of:
2010007H12Rik, 4921523A10Rik, Alox12e, ANKRD52, ANP32C, Cdc14a phosphatase, Cdc14b Phosphatase, LHPP, PP1α, PP1γ, PP2A-δ, PP2A-γ, PP2A-Bα, PP6R3, PPP1R1C, Ppp2r3d, SMEK1, SNED1, SSH3, TBC1D26, Tip41, TM6P1, and TMEM120B.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Fostriecin, 50 µg | sc-202160 | 50 µg | $265.00 |
