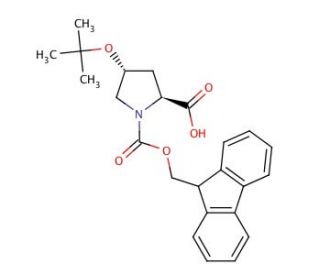
Molecular structure of Fmoc-Hyp(tBu)-OH, CAS Number: 122996-47-8
Fmoc-Hyp(tBu)-OH (CAS 122996-47-8)
Alternate Names:
Fmoc-O-tert-butyl-L-hydroxyproline
CAS Number:
122996-47-8
Molecular Weight:
409.47
Molecular Formula:
C24H27NO5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Fmoc-Hyp(tBu)-OH is a type of amino acid derivative used in peptide synthesis. For example, it has been employed in the synthesis of peptides for use in studies of protein-protein interactions, protein-ligand binding, and enzyme kinetics. Fmoc-Hyp(tBu)-OH acts as a protecting group in peptide synthesis. It is used to protect the amine group of the amino acid from unwanted reactions, such as oxidation and hydrolysis. The tert-butyl group attached to the hydroxyl group also serves to increase the stability of the molecule, allowing it to withstand harsher reaction conditions.
Fmoc-Hyp(tBu)-OH (CAS 122996-47-8) References
- Microwave-assisted synthesis of triple-helical, collagen-mimetic lipopeptides. | Banerjee, J., et al. 2010. Nat Protoc. 5: 39-50. PMID: 20057380
- Interstrand dipole-dipole interactions can stabilize the collagen triple helix. | Shoulders, MD. and Raines, RT. 2011. J Biol Chem. 286: 22905-12. PMID: 21482820
- A 'conovenomic' analysis of the milked venom from the mollusk-hunting cone snail Conus textile--the pharmacological importance of post-translational modifications. | Bergeron, ZL., et al. 2013. Peptides. 49: 145-58. PMID: 24055806
- Non-covalent photo-patterning of gelatin matrices using caged collagen mimetic peptides. | Li, Y., et al. 2015. Macromol Biosci. 15: 52-62. PMID: 25476588
- Chemical Synthesis of a Glycopeptide Derived from Skp1 for Probing Protein Specific Glycosylation. | Chinoy, ZS., et al. 2015. Chemistry. 21: 11779-87. PMID: 26179871
- Nanoparticle Assembly and Gelatin Binding Mediated by Triple Helical Collagen Mimetic Peptide. | San, BH., et al. 2016. ACS Appl Mater Interfaces. 8: 19907-15. PMID: 27403657
- Synthesis and characterization of a photocleavable collagen-like peptide. | Ornelas, A., et al. 2018. Org Biomol Chem. 16: 1000-1013. PMID: 29345707
- Characterization of Hydroxyproline-Containing Hairpin-Like Antimicrobial Peptide EcAMP1-Hyp from Barnyard Grass (Echinochloa crusgalli L.) Seeds: Structural Identification and Comparative Analysis of Antifungal Activity. | Rogozhin, E., et al. 2018. Int J Mol Sci. 19: PMID: 30400225
- Optimization of the Linker Domain in a Dimeric Compound that Degrades an r(CUG) Repeat Expansion in Cells. | Benhamou, RI., et al. 2020. J Med Chem. 63: 7827-7839. PMID: 32657583
- Conjugates of Chitosan and Calcium Alginate with Oligoproline and Oligohydroxyproline Derivatives for Potential Use in Regenerative Medicine. | Wasko, J., et al. 2020. Materials (Basel). 13: PMID: 32664253
- Peptide Probes with Aromatic Residues Tyr and Phe at the X Position Show High Specificity for Targeting Denatured Collagen in Tissues. | Wei, W., et al. 2020. ACS Omega. 5: 33075-33082. PMID: 33403269
- Rational Design of Plant Hairpin-like Peptide EcAMP1: Structural-Functional Correlations to Reveal Antibacterial and Antifungal Activity. | Barashkova, AS., et al. 2022. Molecules. 27: PMID: 35684491
- Chemical Synthesis of Glycopeptides containing l-Arabinosylated Hydroxyproline and Sulfated Tyrosine. | van de Sande, JW. and Albada, B. 2023. Org Lett. 25: 1907-1911. PMID: 36917069
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Fmoc-Hyp(tBu)-OH, 5 g | sc-228190 | 5 g | $276.00 |
