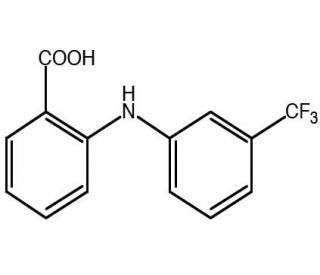
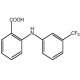
Flufenamic acid (CAS 530-78-9)
See product citations (1)
QUICK LINKS
Flufenamic acid is a nonsteroidal anti-inflammatory compound that functions as an inhibitor of cyclooxygenase, COX-1 and COX-2 enzymes. It exerts its mechanism of action by blocking the production of prostaglandins, which are lipid compounds involved in the inflammatory response. By inhibiting the activity of cyclooxygenase, flufenamic acid interferes with the conversion of arachidonic acid to prostaglandin H2, thereby reducing the synthesis of prostaglandins. At the molecular level, flufenamic acid binds to the active site of the cyclooxygenase enzyme, preventing the conversion of arachidonic acid and subsequent prostaglandin formation. This mechanism of action makes flufenamic acid useful for investigating the role of prostaglandins in various experimental applications, providing insights into the molecular pathways involved in inflammatory processes.
Flufenamic acid (CAS 530-78-9) References
- Fenamates: a novel class of reversible gap junction blockers. | Harks, EG., et al. 2001. J Pharmacol Exp Ther. 298: 1033-41. PMID: 11504800
- Flufenamic acid bi-directionally modulates the transient outward K(+) current in rat cerebellar granule cells. | Zhao, ZG., et al. 2007. J Pharmacol Exp Ther. 322: 195-204. PMID: 17405868
- Flufenamic acid modulates multiple currents in gonadotropin-releasing hormone neurons. | Wang, Y. and Kuehl-Kovarik, MC. 2010. Brain Res. 1353: 94-105. PMID: 20655884
- Flufenamic acid as an ion channel modulator. | Guinamard, R., et al. 2013. Pharmacol Ther. 138: 272-84. PMID: 23356979
- Transdermal iontophoresis of flufenamic acid loaded PLGA nanoparticles. | Malinovskaja-Gomez, K., et al. 2016. Eur J Pharm Sci. 89: 154-62. PMID: 27131608
- Flufenamic acid inhibits osteoclast formation and bone resorption and act against estrogen-dependent bone loss in mice. | Zhang, S., et al. 2020. Int Immunopharmacol. 78: 106014. PMID: 31776093
- Antibacterial and antibiofilm effects of flufenamic acid against methicillin-resistant Staphylococcus aureus. | Zhang, S., et al. 2020. Pharmacol Res. 160: 105067. PMID: 32650057
- Flufenamic Acid-Loaded Self-Nanoemulsifying Drug Delivery System for Oral Delivery: From Formulation Statistical Optimization to Preclinical Anti-Inflammatory Assessment. | Alshehri, S., et al. 2020. J Oleo Sci. 69: 1257-1271. PMID: 32908093
- Targeted delivery of flufenamic acid by V-amylose. | Fatima, R., et al. 2021. Ther Deliv. 12: 575-582. PMID: 34164996
- Engineered Hydrophobin as a Crystallization Inhibitor for Flufenamic Acid. | Sallada, N., et al. 2021. ACS Appl Bio Mater. 4: 6441-6450. PMID: 35006868
- An Osteosarcoma Stem Cell Potent Nickel(II)-Polypyridyl Complex Containing Flufenamic Acid. | Passeri, G., et al. 2022. Molecules. 27: PMID: 35630754
- Flufenamic acid improves survival and neurologic outcome after successful cardiopulmonary resuscitation in mice. | Chen, J., et al. 2022. J Neuroinflammation. 19: 214. PMID: 36050694
- Review of flufenamic acid in rheumatoid arthritis. | Hill, AG. 1966. Ann Phys Med. Suppl: 87-92. PMID: 4895010
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Flufenamic acid, 10 g | sc-205699 | 10 g | $27.00 | |||
Flufenamic acid, 50 g | sc-205699A | 50 g | $79.00 | |||
Flufenamic acid, 100 g | sc-205699B | 100 g | $154.00 | |||
Flufenamic acid, 250 g | sc-205699C | 250 g | $309.00 |
