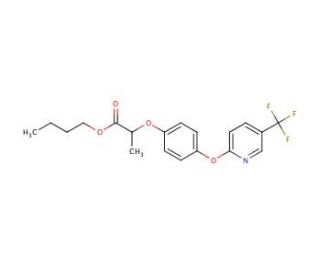
Fluazifop-butyl (CAS 69806-50-4)
QUICK LINKS
Fluazifop-butyl is a selective post-emergence herbicide used extensively in agricultural chemistry research to study weed control, particularly in grassy weed species. The compound belongs to the aryloxyphenoxypropionate group and functions by inhibiting acetyl-CoA carboxylase (ACCase), an enzyme critical for fatty acid biosynthesis in plants. The specificity of Fluazifop-butyl for ACCase in grass species makes it useful for researchers investigating the selective toxicity mechanisms and the development of resistance in target weed populations. Additionally, it is utilized in agronomic studies to assess the impact of herbicide application on crop yield, weed biodiversity, and the environment. Research involving Fluazifop-butyl contributes to the understanding of integrated weed management practices, aiming to optimize the use of herbicides while minimizing negative ecological effects.
Fluazifop-butyl (CAS 69806-50-4) References
- Enantioselective degradation and chiral stability of the herbicide fluazifop-butyl in soil and water. | Qi, Y., et al. 2016. Chemosphere. 146: 315-22. PMID: 26735732
- Enantioselective behaviour of the herbicide fluazifop-butyl in vegetables and soil. | Qi, Y., et al. 2017. Food Chem. 221: 1120-1127. PMID: 27979068
- Ultra-performance convergence chromatography with tandem mass spectrometry-assisted method for rapid enantioseparation and determination of fluazifop-butyl in tobacco and soil. | Yang, F., et al. 2019. Chirality. 31: 353-361. PMID: 30849198
- Effects of the selective herbicide fluazifop on fatty acid synthesis in pea (Pisum sativum) and barley (Hordeum vulgare). | Walker, KA., et al. 1988. Biochem J. 254: 811-7. PMID: 3196294
- Synthesis and application of trifluoromethylpyridines as a key structural motif in active agrochemical and pharmaceutical ingredients. | Tsukamoto, M., et al. 2021. J Pestic Sci. 46: 125-142. PMID: 34135675
- High-performance liquid chromatographic determination of Fluazifop-butyl and Fluazifop in soil and water. | Negre, M., et al. 1987. J Chromatogr. 387: 541-5. PMID: 3558642
- Comparison of two methods for determining the toxicokinetics of fluazifop-butyl after intravenous dosing in rats. | Rawlings, JM., et al. 1994. Hum Exp Toxicol. 13: 123-9. PMID: 7908809
- Effect of dosing vehicle on the dermal absorption of fluazifop-butyl and fomesafen in rats in vivo. | Rawlings, JM., et al. 1994. Fundam Appl Toxicol. 23: 93-100. PMID: 7958570
- The predictive accuracy of in vitro measurements for the dermal absorption of a lipophilic penetrant (fluazifop-butyl) through rat and human skin. | Ramsey, JD., et al. 1994. Fundam Appl Toxicol. 23: 230-6. PMID: 7982531
- Modelling dermal pharmacokinetics using in vitro data. Part I. Fluazifop-butyl in the rat. | Auton, TR., et al. 1993. Hum Exp Toxicol. 12: 199-206. PMID: 8100430
- Microbial metabolism of fluazifop-butyl. | Nègre, M., et al. 1993. J Environ Sci Health B. 28: 545-76. PMID: 8409232
- Fate of fluazifop butyl in rat and human skin in vitro. | Clark, NW., et al. 1993. Arch Toxicol. 67: 44-8. PMID: 8452479
- Effect of diphenyl ether herbicides and oxadiazon on porphyrin biosynthesis in mouse liver, rat primary hepatocyte culture and HepG2 cells. | Krijt, J., et al. 1993. Arch Toxicol. 67: 255-61. PMID: 8517781
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Fluazifop-butyl, 250 mg | sc-235142 | 250 mg | $92.00 |
