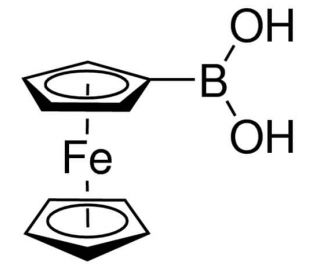
Ferroceneboronic acid (CAS 12152-94-2)
QUICK LINKS
Ferroceneboronic acid is synthesized through a direct reaction between ferrocenyl Grignard reagents and trimethyl borate, followed by hydrolysis. The boronic acid group in ferroceneboronic acid can act as a Lewis acid catalyst in various organic reactions. The ferrocene moiety can potentially stabilize the transition state of the reaction or modify the electronic properties of the boronic acid, affecting the catalytic activity. Researchers investigate the mechanistic aspects of these reactions and the reusability of the ferroceneboronic acid catalyst. The electrochemical properties of ferroceneboronic acid are significant due to the redox-active ferrocene core. Studies focus on the electrochemical behavior of this compound and its potential applications in sensors or electronic devices.
Ferroceneboronic acid (CAS 12152-94-2) References
- Voltammetric response of ferroceneboronic acid to diol and phenolic compounds as possible pollutants. | Takahashi, S., et al. 2011. J Environ Sci (China). 23: 1027-32. PMID: 22066227
- Electrochemical determination of catechin, protocatechuic acid, and L-lactic acid based on voltammetric response of ferroceneboronic acid. | Shi, W., et al. 2014. J AOAC Int. 97: 1742-5. PMID: 25632453
- A sensitive signal-off electrogenerated chemiluminescence biosensing method for the discrimination of DNA hydroxymethylation based on glycosylation modification and signal quenching from ferroceneboronic acid. | Zhang, Y., et al. 2017. Talanta. 170: 546-551. PMID: 28501209
- Synthesis of Water-Dispersed Ferrecene/Phenylboronic Acid-Modified Bifunctional Gold Nanoparticles and the Application in Biosensing. | Xing, Y., et al. 2014. Materials (Basel). 7: 5554-5564. PMID: 28788145
- Spectroscopic insight into supramolecular assemblies of boric acid derivatives and β-cyclodextrin. | Kasprzak, A., et al. 2018. Carbohydr Polym. 198: 294-301. PMID: 30093002
- Redox-Active Dynamic Self-Supporting Thixotropic 3D-Printable G-Quadruplex Hydrogels. | Biswas, A., et al. 2018. Chem Asian J. 13: 3928-3934. PMID: 30334370
- Geodesic-Planar Conjugates: Substituted Buckybowls-Synthesis, Photoluminescence and Electrochemistry. | Bayer, J., et al. 2020. Chemistry. 26: 17546-17558. PMID: 32846003
- Magnetic bead-based electrochemical and colorimetric assays of circulating tumor cells with boronic acid derivatives as the recognition elements and signal probes. | Xia, N., et al. 2021. Talanta. 221: 121640. PMID: 33076160
- Self-healing mechanism and bioelectrochemical interface properties of core-shell guanosine-borate hydrogels. | Wang, H., et al. 2021. J Colloid Interface Sci. 590: 103-113. PMID: 33524710
- Self-Assembly of Functionalized Lipophilic Guanosines into Cation-Free Stacked Guanine-Quartets. | Campitiello, M., et al. 2021. J Org Chem. 86: 9970-9978. PMID: 34279932
- Redox-dependent cytotoxicity of ferrocene derivatives and ROS-activated prodrugs based on ferrocenyliminoboronates. | Věžník, J., et al. 2021. J Inorg Biochem. 224: 111561. PMID: 34385077
- Synthesis, structure and in vitro antiproliferative effects of alkyne-linked 1,2,4-thiadiazole hybrids including erlotinib- and ferrocene-containing derivatives. | Boulhaoua, M., et al. 2021. RSC Adv. 11: 28685-28697. PMID: 35478544
- Ferrocene donor linked to pyridine/pyridinium acceptor via a systematically enlarged π-linker. | Kulhánek, J., et al. 2021. RSC Adv. 11: 38804-38813. PMID: 35493213
- The combination of highly efficient resonance energy transfer in one nanocomposite and ferrocene-quenching for ultrasensitive electrochemiluminescence bioanalysis. | Wang, Y., et al. 2022. Biosens Bioelectron. 210: 114347. PMID: 35550937
- Optimization of Nanohybrid Biosensors Based on Electro-Crosslinked Tannic Acid Capped Nanoparticles/Enzyme. | Savin, R., et al. 2022. Molecules. 27: PMID: 35630787
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ferroceneboronic acid, 1 g | sc-500783 | 1 g | $265.00 |
