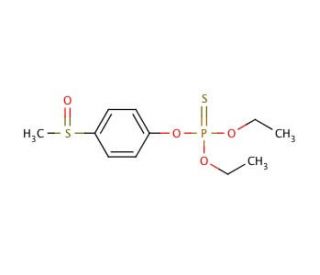
Fensulfothion (CAS 115-90-2)
QUICK LINKS
Fensulfothion is a yellow or brown, oily liquid used as an insecticide, nematocide, and mosquito larvicide, as reported by the EPA in 1998. It falls under the category of organic thiophosphates and sulfoxides and functions as an insecticide inhibiting the EC 3.1.1.7 enzyme (acetylcholinesterase). Additionally, it serves as an agrochemical, avicide, and nematicide, derived from a 4-(methylsulfinyl)phenol. Organophosphorus pesticides, like fensulfothion, exert their toxic effect primarily by inhibiting esterase enzyme activity, especially cholinesterase. These pesticides can also indirectly interact with acetylcholine′s biochemical receptors. When it comes to the reactivation of phosphorylated enzymes, such as acetylated acetylcholinesterase, these esters can be hydrolyzed by nucleophilic agents, including water. However, the rate of reactivation by water for phosphorylated enzymes is extremely low compared to acetylcholinesterase combined with acetate. Inhibition by isopropyl phosphate essentially results in a rate of reactivation close to zero. These findings contribute to the understanding of the mechanisms involved in the toxicity of organic phosphorus pesticides like fensulfothion.
Fensulfothion (CAS 115-90-2) References
- Reduction of fensulfothion to fensulfothion sulfide by Klebsiella pneumoniae. | Wood, PA. and MacRae, IC. 1977. Appl Environ Microbiol. 34: 247-50. PMID: 143907
- Population Dynamics of Belonolaimus longicaudatus and Criconemella ornata and Growth Response of Bermudagrass and Overseeded Grasses on Golf Greens Following Treatment with Nematicides. | Lucas, LT. 1982. J Nematol. 14: 358-63. PMID: 19295721
- Deoxidation of fenthion sulfoxide, fenthion oxon sulfoxide and fensulfothion in gas chromatograph/mass spectrometer, and the prevention of sulfoxide deoxidation by polyethylene glycol 300. | Sugitate, K., et al. 2012. Anal Sci. 28: 669-73. PMID: 22790368
- Oxidative degradation of fensulfothion by hydroxyl radical in aqueous medium. | Sunil Paul, MM., et al. 2013. Chemosphere. 91: 295-301. PMID: 23273737
- Bacterial reduction of fensulfothion and its hydrolysis product 4-methylsulfinyl phenol. | Mac Rae, IC. and Cameron, AJ. 1985. Appl Environ Microbiol. 49: 236-7. PMID: 3156557
- High-Throughput Aptamer Microarrays for Fluorescent Detection of Multiple Organophosphorus Pesticides in Food. | Wang, X., et al. 2022. Anal Chem. 94: 3173-3179. PMID: 35133802
- Degradation of the insecticide fensulfothion by a mixed culture of soil microorganisms. | Miles, JR. and Moy, P. 1982. J Environ Sci Health B. 17: 675-81. PMID: 6220051
- Conversion of fensulfothion by Klebsiella pneumoniae to fensulfothion sulfide and its accumulation. | Timms, P. and MacRae, IC. 1982. Aust J Biol Sci. 35: 661-7. PMID: 6220691
- Reduction of fensulfothion and accumulation of the product, fensulfothion sulfide, by selected microbes. | Timms, P. and MacRae, IC. 1983. Bull Environ Contam Toxicol. 31: 112-5. PMID: 6224524
- Metabolism of fensulfothion by a soil bacterium, Pseudomonas alcaligenes C1. | Sheela, S. and Pai, SB. 1983. Appl Environ Microbiol. 46: 475-9. PMID: 6226243
- Residues of fensulfothion in selected vegetable crops. | Stein, ER., et al. 1980. J Agric Food Chem. 28: 164-7. PMID: 6444643
- Adsorption, desorption, soil mobility and aqueous persistence of fensulfothion and its sulfide and sulfone metabolites. | Miles, JR., et al. 1981. J Environ Sci Health B. 16: 309-24. PMID: 6454719
- Persistence of fensulfothion in a sandy-loam soil and uptake by rutabagas, carrots and radishes using microplots. | Greenhalgh, R. and Read, DC. 1981. J Environ Sci Health B. 16: 363-79. PMID: 6454720
- Poisoning of bald eagles and red-tailed hawks by carbofuran and fensulfothion in the Fraser Delta of British Columbia, Canada. | Elliott, JE., et al. 1996. J Wildl Dis. 32: 486-91. PMID: 8827674
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Fensulfothion, 100 mg | sc-255173 | 100 mg | $93.00 |
