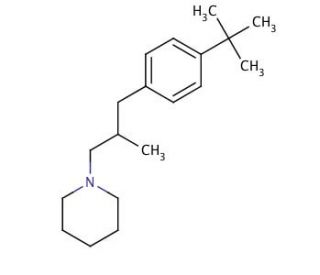
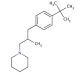
Molecular structure of Fenpropidin, CAS Number: 67306-00-7
Fenpropidin (CAS 67306-00-7)
Application:
Fenpropidin is a fungicide
CAS Number:
67306-00-7
Molecular Weight:
273.46
Molecular Formula:
C19H31N
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Fenpropidin is a fungicide. This compound belongs to the family of piperidines and falls into the category of tertiary amines, where a hydrogen atom in one of the methyl groups is substituted by a p-tert-butylphenyl group. Fenpropidin, belonging to the morpholine class, is an antifungal agent that selectively inhibits the activity of sterol 14-reductase, a enzyme involved in fungal growth and development.
Fenpropidin (CAS 67306-00-7) References
- Construction and growth properties of a yeast strain defective in sterol 14-reductase. | Marcireau, C., et al. 1992. Curr Genet. 22: 267-72. PMID: 1394506
- Molar absorptivities of 2,4-D, cymoxanil, fenpropidin, isoproturon and pyrimethanil in aqueous solution in the near-UV. | Feigenbrugel, V., et al. 2006. Spectrochim Acta A Mol Biomol Spectrosc. 63: 103-10. PMID: 16256424
- Pesticide cocktails can interact synergistically on aquatic crustaceans. | Nørgaard, KB. and Cedergreen, N. 2010. Environ Sci Pollut Res Int. 17: 957-67. PMID: 20077025
- Investigations into the source of two fungicides measured in the air for 24 hours following application to a cereal crop. | Ellis, MC., et al. 2009. Commun Agric Appl Biol Sci. 74: 37-46. PMID: 20218509
- Dissipation and residue of fenpropidin in wheat and soil under field conditions. | Zhao, H., et al. 2012. Ecotoxicol Environ Saf. 77: 52-6. PMID: 22078112
- Silicon Incorporated Morpholine Antifungals: Design, Synthesis, and Biological Evaluation. | Jachak, GR., et al. 2015. ACS Med Chem Lett. 6: 1111-6. PMID: 26617963
- Residues of plant protection products in grey partridge eggs in French cereal ecosystems. | Bro, E., et al. 2016. Environ Sci Pollut Res Int. 23: 9559-73. PMID: 26841780
- Stereoselective Metabolism of the Sterol Biosynthesis Inhibitor Fungicides Fenpropidin, Fenpropimorph, and Spiroxamine in Grapes, Sugar Beets, and Wheat. | Buerge, IJ., et al. 2016. J Agric Food Chem. 64: 5301-9. PMID: 27248479
- Currently and recently used pesticides in Central European arable soils. | Hvězdová, M., et al. 2018. Sci Total Environ. 613-614: 361-370. PMID: 28917175
- Spatial and temporal distribution of the currently-used and recently-banned pesticides in arable soils of the Czech Republic. | Kosubová, P., et al. 2020. Chemosphere. 254: 126902. PMID: 32361539
- Application of the Sea Urchin Embryo Test in Toxicity Evaluation and Effect-Directed Analysis of Wastewater Treatment Plant Effluents. | Mijangos, L., et al. 2020. Environ Sci Technol. 54: 8890-8899. PMID: 32525664
- Gene regulation by morpholines and piperidines in the cardiac embryonic stem cell test. | Mennen, RH., et al. 2021. Toxicol Appl Pharmacol. 433: 115781. PMID: 34737147
- The identification of a gene family in the Saccharomyces cerevisiae ergosterol biosynthesis pathway. | Lai, MH., et al. 1994. Gene. 140: 41-9. PMID: 8125337
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Fenpropidin, 50 mg | sc-235129 | 50 mg | $137.00 |
