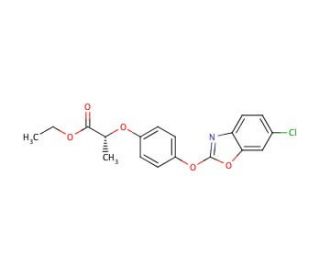
Fenoxaprop-P-ethyl (CAS 71283-80-2)
QUICK LINKS
Fenoxaprop-P-ethyl, a herbicide widely employed in agricultural fields and gardens, effectively tackles the issue of weed control. Belonging to the aryloxyphenoxypropionic acid (AOPP) family, a group of herbicides renowned for inhibiting weed growth, Fenoxaprop-P-ethyl showcases selective properties, targeting specific weed species while leaving others unaffected. Its efficacy is further enhanced when combined with other herbicides, offering comprehensive weed control. The mechanism of action of Fenoxaprop-P-ethyl lies in the inhibition of acetolactate synthase (ALS), a pivotal enzyme involved in the biosynthesis of branched-chain amino acids. Specifically, it binds to the active site of the enzyme, impeding its catalytic ability to facilitate the production of branched-chain amino acids. Consequently, the reduced availability of these amino acids hampers plant growth, ultimately leading to the demise of the targeted plants.
Fenoxaprop-P-ethyl (CAS 71283-80-2) References
- Biodegradation of fenoxaprop-p-ethyl by bacteria isolated from sludge. | Song, L., et al. 2005. J Hazard Mater. 118: 247-51. PMID: 15721550
- Residues and dissipation of the herbicide fenoxaprop-P-ethyl and its metabolite in wheat and soil. | Chen, X., et al. 2011. Bull Environ Contam Toxicol. 87: 50-3. PMID: 21533830
- Identification and mechanism of Echinochloa crus-galli resistance to fenoxaprop-p-ethyl with respect to physiological and anatomical differences. | Hamza, A., et al. 2012. ScientificWorldJournal. 2012: 893204. PMID: 22666165
- The role of cytochrome P450 monooxygenase in the different responses to fenoxaprop-P-ethyl in annual bluegrass (Poa annua L.) and short awned foxtail (Alopecurus aequalis Sobol.). | Wang, H., et al. 2013. Pestic Biochem Physiol. 107: 334-42. PMID: 24267695
- miR397/Laccase Gene Mediated Network Improves Tolerance to Fenoxaprop-P-ethyl in Beckmannia syzigachne and Oryza sativa. | Pan, L., et al. 2017. Front Plant Sci. 8: 879. PMID: 28588605
- Fenoxaprop-P-ethyl resistance conferred by cytochrome P450s and target site mutation in Alopecurus japonicus. | Chen, G., et al. 2018. Pest Manag Sci. 74: 1694-1703. PMID: 29337421
- Target site mutations and cytochrome P450s confer resistance to fenoxaprop-P-ethyl and mesosulfuron-methyl in Alopecurus aequalis. | Zhao, N., et al. 2019. Pest Manag Sci. 75: 204-214. PMID: 29808621
- Fenoxaprop-P-ethyl and mesosulfuron-methyl resistance status of shortawn foxtail (Alopecurus aequalis Sobol.) in eastern China. | Guo, W., et al. 2018. Pestic Biochem Physiol. 148: 126-132. PMID: 29891363
- Trp-1999-Ser mutation of acetyl-CoA carboxylase and cytochrome P450s-involved metabolism confer resistance to fenoxaprop-P-ethyl in Polypogon fugax. | Zhao, N., et al. 2019. Pest Manag Sci. 75: 3175-3183. PMID: 30950550
- Degradation of Fenoxaprop-p-Ethyl and Its Metabolite in Soil and Wheat Crops. | Tandon, S. 2019. J Food Prot. 82: 1959-1964. PMID: 31638411
- Identification and expression of main genes involved in non-target site resistance mechanisms to fenoxaprop-p-ethyl in Beckmannia syzigachne. | Bai, S., et al. 2020. Pest Manag Sci. 76: 2619-2626. PMID: 32083373
- Identification of essential genes involved in metabolism-based resistance mechanism to fenoxaprop-P-ethyl in Polypogon fugax. | Zhao, N., et al. 2022. Pest Manag Sci. 78: 1164-1175. PMID: 34821014
- The safener isoxadifen-ethyl confers fenoxaprop-p-ethyl resistance on a biotype of Echinochloa crus-galli. | Cutti, L., et al. 2022. Pest Manag Sci. 78: 2287-2298. PMID: 35220677
- Transcriptome analysis and the identification of genes involved in the metabolic pathways of fenoxaprop-P-ethyl in rice treated with isoxadifen-ethyl hydrolysate. | Zhao, Y., et al. 2022. Pestic Biochem Physiol. 183: 105057. PMID: 35430061
- Degradation dynamics, correlations, and residues of carfentrazone-ethyl, fenoxaprop-p-ethyl, and pinoxaden under the continuous application in the wheat field. | Sondhia, S., et al. 2023. Environ Geochem Health.. PMID: 36700995
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Fenoxaprop-P-ethyl, 250 mg | sc-235126 | 250 mg | $84.00 |
