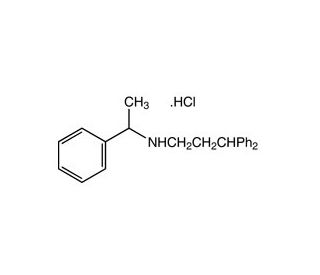
Fendiline hydrochloride (CAS 13636-18-5)
See product citations (3)
QUICK LINKS
Fendiline hydrochloride, a powerful and specific agonist, displays a remarkable affinity for the dopamine D2 receptor. By binding to this receptor, it initiates a chain of events that culminate in the release of dopamine—a pivotal neurotransmitter. The activation of the dopamine D2 receptor sets in motion a cascade of effects that extend beyond the immediate receptor itself. This includes the activation of other receptors, the subsequent release of additional neurotransmitters, and the induction of diverse cellular processes. The intricate interplay triggered by Fendiline hydrochloride′s interaction with the dopamine D2 receptor holds significant implications for neurotransmission and cellular signaling. Its potent agonistic properties render it a notable subject of investigation in the realms of neuroscience and pharmacology.
Fendiline hydrochloride (CAS 13636-18-5) References
- [Coronary collateral vessels after long-term treatment with fendiline hydrochloride in a double-blind study (author's transl)]. | Knoche, H. and Schmitt, G. 1975. Arzneimittelforschung. 25: 1781-2. PMID: 1106442
- Inhibitors of cytochrome c release with therapeutic potential for Huntington's disease. | Wang, X., et al. 2008. J Neurosci. 28: 9473-85. PMID: 18799679
- Clinically applicable antianginal agents suppress osteoblastic transformation of myogenic cells and heterotopic ossifications in mice. | Yamamoto, R., et al. 2013. J Bone Miner Metab. 31: 26-33. PMID: 23011467
- Perhexiline maleate enhances antitumor efficacy of cisplatin in neuroblastoma by inducing over-expression of NDM29 ncRNA. | Vella, S., et al. 2015. Sci Rep. 5: 18144. PMID: 26674674
- Novel cell-based in vitro screen to identify small-molecule inhibitors against intracellular replication of Cryptococcus neoformans in macrophages. | Samantaray, S., et al. 2016. Int J Antimicrob Agents. 48: 69-77. PMID: 27289450
- Co-Administration of Fendiline Hydrochloride Enhances Chemotherapeutic Efficacy of Cisplatin in Neuroblastoma Treatment. | Brizzolara, A., et al. 2020. Molecules. 25: PMID: 33182713
- Identification of Potent and Safe Antiviral Therapeutic Candidates Against SARS-CoV-2. | Xiao, X., et al. 2020. Front Immunol. 11: 586572. PMID: 33324406
- Efficacy of a Three Drug-Based Therapy for Neuroblastoma in Mice. | Garbati, P., et al. 2021. Int J Mol Sci. 22: PMID: 34201814
- Mechanisms of hedgehog, calcium and retinoic acid signalling pathway inhibitors: Plausible modes of action along the MLL-EZH2-p53 axis in cellular growth control. | Manna, S., et al. 2023. Arch Biochem Biophys. 742: 109600. PMID: 37142078
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Fendiline hydrochloride, 5 g | sc-239988 | 5 g | $119.00 |
