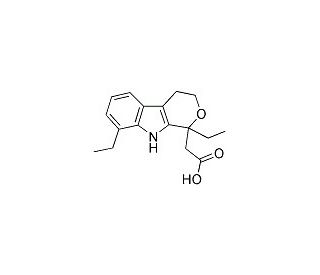
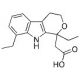
Etodolac (CAS 41340-25-4)
See product citations (1)
QUICK LINKS
Etodolac is a nonsteroidal anti-inflammatory compound that functions as an inhibitor of the enzyme cyclooxygenase (COX). It specifically inhibits the COX-2 enzyme, which is responsible for the production of prostaglandins involved in inflammation and pain. By blocking the action of COX-2, etodolac reduces the synthesis of prostaglandins, thereby decreasing inflammation and pain. This chemical′s mode of action involves binding to the active site of the COX-2 enzyme, preventing the conversion of arachidonic acid into prostaglandins. This inhibition of prostaglandin synthesis contributes to the anti-inflammatory and analgesic effects of etodolac. Additionally, etodolac may act to have some selectivity for COX-2 over COX-1, which may contribute to its reduced gastrointestinal side effects compared to other NSAIDs. Overall, etodolac′s function as a COX-2 inhibitor makes it a useful tool for studying the role of prostaglandins in inflammation and pain pathways at the molecular level.
Etodolac (CAS 41340-25-4) References
- Etodolac inhibits EBER expression and induces Bcl-2-regulated apoptosis in Burkitt's lymphoma cells. | Kobayashi, M., et al. 2005. Eur J Haematol. 75: 212-20. PMID: 16104877
- Etodolac. A reappraisal of its pharmacology and therapeutic use in rheumatic diseases and pain states. | Balfour, JA. and Buckley, MM. 1991. Drugs. 42: 274-99. PMID: 1717225
- Etodolac attenuates mechanical allodynia in a mouse model of neuropathic pain. | Inoue, N., et al. 2009. J Pharmacol Sci. 109: 600-5. PMID: 19346674
- Etodolac: efficacy in osteoarthritis and effects on chondrocyte function. | Bacon, PA. 1990. Rheumatol Int. 10 Suppl: 3-7. PMID: 2150570
- Etodolac blocks the allyl isothiocyanate-induced response in mouse sensory neurons by selective TRPA1 activation. | Inoue, N., et al. 2012. Pharmacology. 90: 47-54. PMID: 22739568
- Etodolac activates and desensitizes transient receptor potential ankyrin 1. | Wang, S., et al. 2013. J Neurosci Res. 91: 1591-8. PMID: 24027177
- Etodolac. A preliminary review of its pharmacodynamic activity and therapeutic use. | Lynch, S. and Brogden, RN. 1986. Drugs. 31: 288-300. PMID: 2940079
- Pharmacokinetic modeling and simulation of etodolac following single oral administration in dogs. | Baek, IH. 2019. Xenobiotica. 49: 981-986. PMID: 30216103
- Improved degradation of etodolac in the presence of core-shell ZnFe2O4/SiO2/TiO2 magnetic photocatalyst. | Mrotek, E., et al. 2020. Sci Total Environ. 724: 138167. PMID: 32251886
- Transfersome Hydrogel Containing 5-Fluorouracil and Etodolac Combination for Synergistic Oral Cancer Treatment. | Bollareddy, SR., et al. 2022. AAPS PharmSciTech. 23: 70. PMID: 35132496
- Etodolac Enhances the Radiosensitivity of Irradiated HT-29 Human Colorectal Cancer Cells. | Shaghaghi, Z., et al. 2022. Curr Radiopharm. 15: 242-248. PMID: 35319403
- Cardiovascular Risks of Diclofenac Versus Other Older COX-2 Inhibitors (Meloxicam and Etodolac) and Newer COX-2 Inhibitors (Celecoxib and Etoricoxib): A Series of Nationwide Emulated Trials. | Schmidt, M., et al. 2022. Drug Saf. 45: 983-994. PMID: 35909207
- Application of ionic liquid to enhance the nose-to-brain delivery of etodolac. | Tanigawa, H., et al. 2022. Eur J Pharm Sci. 178: 106290. PMID: 36058500
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Etodolac, 100 mg | sc-204747 | 100 mg | $89.00 | |||
| US: (Out of Stock: Availability February 11, 2026) | ||||||
Etodolac, 250 mg | sc-204747A | 250 mg | $131.00 | |||
