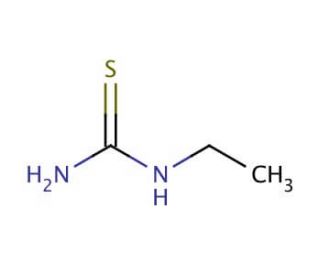
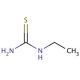
Ethylthiourea (CAS 625-53-6)
QUICK LINKS
Ethylthiourea is an organosulfur compound extensively used in research to study its diverse biological activities and chemical properties. Its mechanism of action primarily involves the inhibition of enzyme activity through its interaction with thiol groups, which can lead to the disruption of various biochemical pathways. Ethylthiourea has been employed in studies investigating its role as an inhibitor of tyrosinase, an enzyme critical in melanin synthesis, making it a valuable tool in research on pigmentation and enzyme regulation. Additionally, it has been utilized in the synthesis of other sulfur-containing compounds and as a ligand in coordination chemistry, aiding in the exploration of metal complexes with potential catalytic properties. Recent research has focused on the environmental impact and degradation pathways of ethylthiourea, examining its persistence and transformation in different ecosystems. Advanced analytical techniques such as mass spectrometry and nuclear magnetic resonance (NMR) spectroscopy have been used to characterize its interactions and stability. Moreover, ethylthiourea serves as a model compound in toxicological studies to understand the effects of thiourea derivatives on biological systems, providing insights into their mechanisms of toxicity and potential environmental hazards. Its multifaceted applications highlight its significance in chemical, biological, and environmental research.
Ethylthiourea (CAS 625-53-6) References
- Novel thiourea compounds as dual-function microbicides. | D'Cruz, OJ., et al. 2000. Biol Reprod. 63: 196-205. PMID: 10859260
- Infrared spectra of new Re(III) complexes with thiourea derivatives. | Gambino, D., et al. 2002. Spectrochim Acta A Mol Biomol Spectrosc. 58: 3085-92. PMID: 12511092
- Conformational preferences and internal rotation in alkyl- and phenyl-substituted thiourea derivatives. | Bryantsev, VS. and Hay, BP. 2006. J Phys Chem A. 110: 4678-88. PMID: 16599434
- Synthesis and cytostatic properties of daunorubicin derivatives, containing N-phenylthiourea or N-ethylthiourea moieties in the 3'-position. | Preobrazhenskaya, MN., et al. 1991. J Antibiot (Tokyo). 44: 192-9. PMID: 1901311
- Unveiling first report on in silico modeling of aquatic toxicity of organic chemicals to Labeo rohita (Rohu) employing QSAR and q-RASAR. | Gallagher, A. and Kar, S. 2024. Chemosphere. 349: 140810. PMID: 38029938
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethylthiourea, 10 g | sc-203333 | 10 g | $138.00 |
