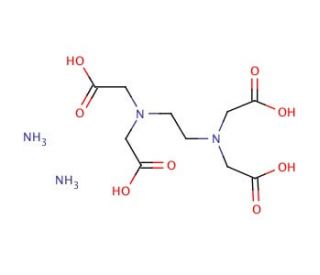
Ethylenediaminetetraacetic acid diammonium salt (CAS 20824-56-0)
QUICK LINKS
Ethylenediaminetetraacetic acid diammonium salt (EDTA diammonium salt) is a widely utilized chelating agent in various research fields due to its ability to form stable complexes with metal ions. In analytical chemistry, Ethylenediaminetetraacetic acid diammonium salt is employed extensively in complexometric titrations, a method used to determine the concentration of metal ions in a solution. Its strong chelating ability allows for the precise measurement of metals by forming stable, non-reactive complexes. This property is for the accurate quantification of metal ions in water samples, pharmaceuticals, and food products. Ethylenediaminetetraacetic acid diammonium salt plays a vital role in biochemistry and molecular biology labs. It is used to chelate divalent cations such as Mg²⁺ and Ca²⁺, which are essential cofactors for enzymatic activities including DNases and RNases. By binding these metal ions, Ethylenediaminetetraacetic acid diammonium salt inhibits these enzymes and prevents the degradation of nucleic acids during extraction and purification processes. It is also a component of various buffers used in DNA and RNA extraction protocols, as well as in polymerase chain reactions (PCR), where it helps maintain the integrity of nucleic acids. In environmental studies, Ethylenediaminetetraacetic acid diammonium salt is used for the remediation of heavy metal-contaminated soils and waters. Its chelating properties allow it to bind with heavy metals, making them more soluble and easier to remove or recover from the environment. This application is significant for cleaning up industrial waste and mitigating pollution.
Ethylenediaminetetraacetic acid diammonium salt (CAS 20824-56-0) References
- Donor-donor energy-migration measurements of dimeric DsbC labeled at its N-terminal amines with fluorescent probes: a study of protein unfolding. | Duan, X., et al. 2004. Angew Chem Int Ed Engl. 43: 4216-9. PMID: 15307091
- Systemic lanthanum is excreted in the bile of rats. | Damment, SJ. and Pennick, M. 2007. Toxicol Lett. 171: 69-77. PMID: 17570622
- Rapid metal extractability tests from polluted mining soils by ultrasound probe sonication and microwave-assisted extraction systems. | García-Salgado, S. and Quijano, MÁ. 2016. Environ Sci Pollut Res Int. 23: 24567-24577. PMID: 27826824
- Physiochemical characterization and systematic investigation of metals extraction from fly and bottom ashes produced from municipal solid waste. | Al-Ghouti, MA., et al. 2020. PLoS One. 15: e0239412. PMID: 33091005
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethylenediaminetetraacetic acid diammonium salt, 500 g | sc-215006 | 500 g | $302.00 |
