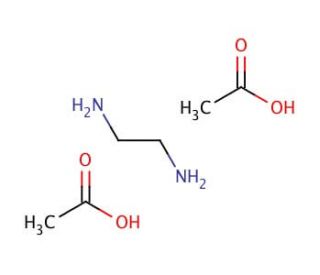
Ethylenediamine diacetate (CAS 38734-69-9)
QUICK LINKS
Ethylenediamine diacetate is a versatile compound widely utilized in various research applications, particularly for its chelating properties. This compound acts by forming complex coordination bonds with metal ions, which can significantly alter the reactivity, solubility, and stability of metal-containing systems. Its mechanism of action involves the ethylenediamine moiety providing a flexible backbone that can adjust its conformation to optimally coordinate with metal ions, while the acetate groups serve to enhance solubility and further stabilize metal complexes. This dual functionality makes it a compound of interest in the synthesis and modification of metal-organic frameworks (MOFs), catalysis research, and the study of metal-mediated processes in environmental science. In addition to its role in binding and stabilizing metal ions, ethylenediamine diacetate is explored for its potential in facilitating organic transformations, serving as a ligand in catalytic systems, and acting as a key component in the development of novel materials with specific electronic or magnetic properties.
Ethylenediamine diacetate (CAS 38734-69-9) References
- Concise total synthesis of biologically interesting mallotophilippens C and e. | Lee, YR., et al. 2008. J Org Chem. 73: 4313-6. PMID: 18442292
- Metal-free Brønsted acid catalyzed formal [3 + 3] annulation. straightforward synthesis of dihydro-2H-chromenones, pyranones, and tetrahydroquinolinones. | Moreau, J., et al. 2009. J Org Chem. 74: 8963-73. PMID: 19950880
- Ethylenediamine diacetate (EDDA) mediated synthesis of aurones under ultrasound: their evaluation as inhibitors of SIRT1. | Manjulatha, K., et al. 2012. Bioorg Med Chem Lett. 22: 6160-5. PMID: 22929231
- Efficient one-step synthesis of pyrrolo[3,4-c]quinoline-1,3-dione derivatives by organocatalytic cascade reactions of isatins and β-ketoamides. | Xia, L. and Lee, YR. 2013. Org Biomol Chem. 11: 5254-63. PMID: 23824233
- Microwave-Assisted Organocatalytic Intramolecular Knoevenagel/Hetero Diels-Alder Reaction with O-(Arylpropynyloxy)-Salicylaldehydes: Synthesis of Polycyclic Embelin Derivatives. | Martín-Acosta, P., et al. 2016. J Org Chem. 81: 9738-9756. PMID: 27680299
- Efficient and general method for the synthesis of benzopyrans by ethylenediamine diacetate-catalyzed reactions of resorcinols with α,β-unsaturated aldehydes. One step synthesis of biologically active (±)-confluentin and (±)-daurichromenic acid | Yong Rok Lee, Jung Hyun Choi, Sang Heum Yoon. 2005. Tetrahedron Letters. 46: 7539-7543.
- A Novel Method for the Synthesis of Substituted Benzochromenes by Ethylenediamine Diacetate-Catalyzed Cyclizations of Naphthalenols to α,β-Unsaturated Aldehydes. Concise Synthesis of the Natural Products Lapachenole, Dihydrolapachenole, and Mollugin | Yong Rok Lee, Yun Mi Kim. 2007. Helvetica Chimica Acta. 90: 2401-2413.
- Ethylenediamine diacetate (EDDA)-catalyzed one-pot synthesis of tetrahydroquinolines by domino Knoevenagel/hetero Diels–Alder reactions from 1,3-dicarbonyls | Yong Rok Lee, Thai Viet Hung. 2008. Tetrahedron. 64: 7338-7346.
- Efficient one-pot synthetic approaches for cannabinoid analogues and their application to biologically interesting (−)-hexahydrocannabinol and (+)-hexahydrocannabinol | Yong Rok Lee, Likai Xia. 2008. Tetrahedron Letters. 49: 3283-3287.
- Ethylenediamine diacetate-catalyzed three-component reaction for the synthesis of 2,3-dihydroquinazolin-4(1H)-ones and their spirooxindole derivatives | Manchala Narasimhulu, Yong Rok Lee. 2011. Tetrahedron. 67: 9627-9634.
- Synthesis of new chromeno-annulated cis-fused pyrano[3,4-c]pyran derivatives via domino Knoevenagel–hetero-Diels–Alder reactions and their biological evaluation towards antiproliferative activity | A. Venkatesham,a R. Srinivasa Rao,a K. Nagaiah,*a J. S. Yadav,a G. RoopaJones,b S. J. Basha,c B. Sridhard and A. Addlagattab. 2012. Med. Chem. Commun. 3: 652-658.
- A new route to the versatile synthesis of thiopyrano[2,3-b:6,5-b′]diindoles via 2-(alkylthio)-indole-3-carbaldehydes | Mukund Jha, Michael Edmunds, Kate-lyn Lund, Ashley Ryan. 2014. Tetrahedron Letters. 55: 5691-5694.
- Efficient One-Pot Synthesis of Tetrahydrobenzo[b]pyrans by Ethylenediamine Diacetate-Catalyzed Multicomponent Reaction Under Solvent-Free Conditions | Zhongqiang Zhou, Yuliang Zhang & Xiaoyun Hu. 2017. Polycyclic Aromatic Compounds. 37: 39-45.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethylenediamine diacetate, 5 g | sc-252809 | 5 g | $166.00 |
