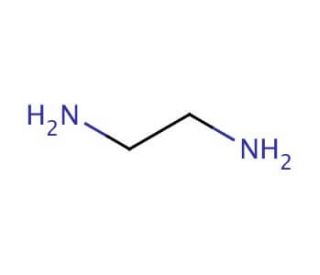
Molecular structure of Ethylenediamine, CAS Number: 107-15-3
Ethylenediamine (CAS 107-15-3)
See product citations (1)
Alternate Names:
1,2-Ethanediamine; 1,2-Diaminoethane
Application:
Ethylenediamine is used as fuel additives, bleach activators, chelating agents, and corrosion inhibitors
CAS Number:
107-15-3
Molecular Weight:
60.10
Molecular Formula:
C2H8N2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Ethylenediamine functions as a chelating agent in research and development applications. It acts by forming coordination complexes with metal ions, which can be useful in various experimental processes such as catalysis, metal extraction, and metal analysis. Ethylenediamine′s mechanism of action involves its ability to bind to metal ions through its two amine groups, forming stable complexes that can be utilized in a range of chemical reactions and analytical techniques. In experimental applications, Ethylenediamine′s role as a chelating agent allows for the manipulation and control of metal ions, contributing to the advancement of various scientific investigations and applications.
Ethylenediamine (CAS 107-15-3) References
- Ethylenediamine as eluent component in cation chromatography. Predictive and comparative study for analysis of alkaline earth ions. | Hajós, P. 2002. J Chromatogr A. 955: 1-8. PMID: 12061555
- Aminophylline hypersensitivity apparently due to ethylenediamine. | Terzian, CG. and Simon, PA. 1992. Ann Emerg Med. 21: 312-4. PMID: 1536494
- Effect of ethylenediamine on chemical degradation of insulin aspart in pharmaceutical solutions. | Poulsen, C., et al. 2008. Pharm Res. 25: 2534-44. PMID: 18607693
- Synthesis, characterization and properties of ethylenediamine-functionalized Fe3O4 magnetic polymers for removal of Cr(VI) in wastewater. | Zhao, YG., et al. 2010. J Hazard Mater. 182: 295-302. PMID: 20621418
- Ethylenediamine: an effective reagent for deacetylation of natural products. | Mohankumar, R., et al. 2010. J Asian Nat Prod Res. 12: 851-8. PMID: 20924898
- Ethylenediamine-induced late asthmatic responses. | Nakazawa, T. and Matsui, S. 1990. J Asthma. 27: 207-12. PMID: 2211492
- Synthesis of ethylenediamine modified chitosan and evaluation for removal of divalent metal ions. | Chethan, PD. and Vishalakshi, B. 2013. Carbohydr Polym. 97: 530-6. PMID: 23911481
- Ethylenediamine-modified oriented MCM-41 at the electrode surface, cobalt adsorption ability and electrochemical performance. | Rafiee, M., et al. 2014. Dalton Trans. 43: 4901-8. PMID: 24492250
- Synthesis of ethylenediamine modified chitosan microspheres for removal of divalent and hexavalent ions. | Chethan, PD. and Vishalakshi, B. 2015. Int J Biol Macromol. 75: 179-85. PMID: 25655019
- Enhancing adsorption of U(VI) onto EDTA modified L. cylindrica using epichlorohydrin and ethylenediamine as a bridge. | Su, S., et al. 2017. Sci Rep. 7: 44156. PMID: 28272435
- Palladium Probe Consisting of Naphthalimide and Ethylenediamine for Selective Turn-On Sensing of CO and Cell Imaging. | Tikum, AF., et al. 2021. Inorg Chem. 60: 7108-7114. PMID: 33904727
- Ethylenediamine as a specific releasing agent of gamma-aminobutyric acid in rat striatal slices. | Lloyd, HG., et al. 1982. J Neurochem. 38: 1168-9. PMID: 7082461
- The use of ethylenediamine in softening hard plant structures for paraffin sectioning. | Carlquist, S. 1982. Stain Technol. 57: 311-7. PMID: 7179333
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethylenediamine, 250 ml | sc-215004 | 250 ml | $36.00 | |||
Ethylenediamine, 1 L | sc-215004A | 1 L | $83.00 |
