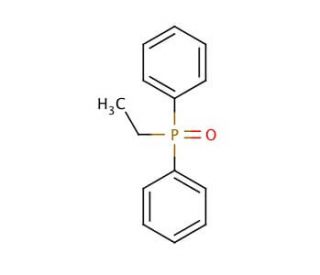
Ethyldiphenylphosphine oxide (CAS 1733-57-9)
QUICK LINKS
Ethyldiphenylphosphine oxide is a specialized organophosphorus compound with potential as a ligand in coordination chemistry. Its structure, featuring both alkyl and aryl groups attached to phosphorus, allows it to coordinate with various metal centers, influencing the reactivity and stability of metal complexes. This coordination is central to studies on catalysis, where ethyldiphenylphosphine oxide-modified catalysts are examined for their efficiency in facilitating reactions such as hydrogenation, carbon-carbon bond formation, and oxidation processes. The compound′s oxide moiety also makes it a point of study in understanding the role of oxygen-containing ligands in altering the electronic properties of metal centers, which is for fine-tuning catalytic activity. Furthermore, in materials science, it has potential applications in the development of new materials with specific optical and electronic properties, as the phosphine oxide group can interact with polymers and other substrates to enhance material performance.
Ethyldiphenylphosphine oxide (CAS 1733-57-9) References
- Catalytic, enantioselective, intramolecular carbosulfenylation of olefins. Preparative and stereochemical aspects. | Denmark, SE. and Jaunet, A. 2014. J Org Chem. 79: 140-71. PMID: 24328051
- Alkenylation of C(sp3)-H Bonds by Zincation/Copper-Catalyzed Cross-Coupling with Iodonium Salts. | Liu, C. and Wang, Q. 2018. Angew Chem Int Ed Engl. 57: 4727-4731. PMID: 29479782
- Interfacial behaviour of diphenylalkylphosphine oxides at the mercury-methanolic solution interface | P Nikitas, A Pappa-Louisi, D Jannakoudakis. 1984. Journal of Electroanalytical Chemistry and Interfacial Electrochemistry. 162(1–2): 175-187.
- Preparation and reactivity of monomeric (tolylimido) molybdenum (V) complexes and structure of mer-trichloro-trans-bis (ethyldiphenylphosphine)(p-tolylimido) molybdenum (V), Mo (Ntol) Cl3 (EtPh2P) 2 | Chou, C. Y., Huffman, J. C., & Maatta, E. A. 1986. Inorganic Chemistry. 25(6): 822-826.
- Synthesis of phenylalanine-derived β-hydroxy and β-keto phosphine oxides-investigation of the configurational stability of lithiated phosphine oxides using the hoffmann test | O'Brien, P., & Warren, S. 1996. Tetrahedron letters. 37(24): 4271-4274.
- Alane - A chemoselective way to reduce phosphine oxides | A Bootle-Wilbraham, S Head, J Longstaff, P Wyatt. 1999. Tetrahedron Letters. 40(28): 5267-5270.
- A study of the behaviour of Cu6(TePh)6(PPh2Et)5 and related phosphine (PPh2Et) and phosphine oxide (Ph2EtPO) adsorbed in mesoporous molecular sieves | CM Kowalchuk, JF Corrigan, Y Huang. 2005. Microporous and Mesoporous Materials. 81(1–3): 211-216.
- Regioselective functionalisation of nitrobenzene and benzonitrile derivatives via nucleophilic aromatic substitution of hydrogen by phosphorus-stabilized carbanions | CMA Sánchez, MJ Iglesias, JG Lopez, IJP Álvarez. 2006. Tetrahedron. 62(15): 3648-3662.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyldiphenylphosphine oxide, 5 g | sc-228086 | 5 g | $293.00 |
