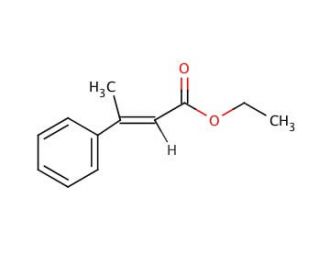
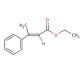
Molecular structure of Ethyl trans-β-methylcinnamate, CAS Number: 1504-72-9
Ethyl trans-β-methylcinnamate (CAS 1504-72-9)
Application:
Ethyl trans-β-methylcinnamate is used in diastereoselective synthesis of ethyl (E)-3-methyl-3-phenylglycidate
CAS Number:
1504-72-9
Molecular Weight:
190.24
Molecular Formula:
C12H14O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Ethyl trans-β-methylcinnamate was used in diastereoselective synthesis of ethyl (E)-3-methyl-3-phenylglycidate. Ethyl trans-β-methylcinnamate is an organic compound known for its volatility. Its versatile nature allows it to serve as a substrate in the investigation of enzyme kinetics and as a model compound for studying various chemical reactions. Additionally, it has found utility as a reference compound for determining the boiling point of other substances.
Ethyl trans-β-methylcinnamate (CAS 1504-72-9) References
- Stereocontrolled IMDA reaction of styrene derivatives. A way to enantiopure 3a,4,9,9a-tetrahydrobenz[f]isoindolines. | Pedrosa, R., et al. 2002. J Org Chem. 67: 782-9. PMID: 11856020
- Conjugate reduction of alpha,beta-unsaturated carbonyl compounds catalyzed by a copper carbene complex. | Jurkauskas, V., et al. 2003. Org Lett. 5: 2417-20. PMID: 12841744
- Ruthenium-catalyzed Heck-type olefination and Suzuki coupling reactions: studies on the nature of catalytic species. | Na, Y., et al. 2004. J Am Chem Soc. 126: 250-8. PMID: 14709090
- Catalytic enantioselective conjugate reduction of lactones and lactams. | Hughes, G., et al. 2003. J Am Chem Soc. 125: 11253-8. PMID: 16220945
- m-Carborane-based chiral NBN pincer-metal complexes: synthesis, structure, and application in asymmetric catalysis. | El-Zaria, ME., et al. 2011. Inorg Chem. 50: 4149-61. PMID: 21438543
- Tandem diastereo- and enantioselective preparation of aryl and alkyl cyclopropyl carbinols with three adjacent stereocenters using perhydrobenzoxazines and diethylzinc. | Infante, R., et al. 2014. Org Biomol Chem. 12: 345-54. PMID: 24264695
- Stereospecific S(N)2@P reactions: novel access to bulky P-stereogenic ligands. | Orgué, S., et al. 2015. Chem Commun (Camb). 51: 17548-51. PMID: 26477668
- Pd nanoparticles on green support as dip-catalyst: a facile transfer hydrogenation of olefins and N-heteroarenes in water. | Shaikh, MN. 2019. RSC Adv. 9: 28199-28206. PMID: 35530451
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl trans-β-methylcinnamate, 5 g | sc-228079 | 5 g | $125.00 |
