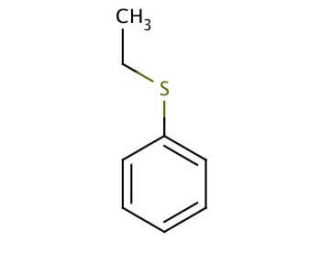
Ethyl phenyl sulfide (CAS 622-38-8)
QUICK LINKS
Ethyl phenyl sulfide is a chemical compound that functions as a nucleophilic reagent in organic synthesis. It participates in substitution reactions, in the formation of carbon-sulfur bonds. Ethyl phenyl sulfide acts as a sulfur source in the synthesis of various organic molecules, serving as a key building block in the construction of sulfur-containing compounds. Ethyl phenyl sulfide undergoes nucleophilic substitution reactions with electrophiles, leading to the formation of new carbon-sulfur bonds. Ethyl phenyl sulfide′s mechanism of action involves the attack of the sulfur atom on electrophilic carbon centers, resulting in the displacement of the leaving group and the formation of the desired carbon-sulfur bond. In this way, ethyl phenyl sulfide plays a role in the construction of complex organic molecules by facilitating the introduction of sulfur atoms at specific positions within the molecular framework.
Ethyl phenyl sulfide (CAS 622-38-8) References
- Dibenzyl sulfide metabolism by white rot fungi. | Van Hamme, JD., et al. 2003. Appl Environ Microbiol. 69: 1320-4. PMID: 12571066
- Biphenyl derived Schiff-base vanadium(V) complexes with pendant OH-groups--structure, characterization and hydrogen peroxide mediated sulfide oxygenation. | Plitt, P., et al. 2004. Dalton Trans. 2314-20. PMID: 15278124
- Discovery of a novel styrene monooxygenase originating from the metagenome. | van Hellemond, EW., et al. 2007. Appl Environ Microbiol. 73: 5832-9. PMID: 17644649
- Degradation of organic sulfur compounds by a coal-solubilizing fungus. | Faison, BD., et al. 1991. Appl Biochem Biotechnol. 28-29: 237-51. PMID: 1929365
- Facile C-S, S-H, and S-S bond cleavage using a nickel(0) NHC complex. | Schaub, T., et al. 2009. Dalton Trans. 7071-9. PMID: 20449150
- Mutations of an NAD(P)H-dependent flavoprotein monooxygenase that influence cofactor promiscuity and enantioselectivity. | Jensen, CN., et al. 2013. FEBS Open Bio. 3: 473-8. PMID: 24251114
- Preparation, Characterization, and Oxygenase Activity of a Photocatalytic Artificial Enzyme. | Gu, Y., et al. 2015. Chembiochem. 16: 1880-1883. PMID: 26097041
- Enzymatic potential of heterotrophic bacteria from a neutral copper mine drainage. | Costa, BZ., et al. 2016. Braz J Microbiol. 47: 846-852. PMID: 27522533
- Labelling and determination of the energy in reactive intermediates in solution enabled by energy-dependent reaction selectivity. | Kurouchi, H. and Singleton, DA. 2018. Nat Chem. 10: 237-241. PMID: 29359761
- Highly Selective Oxidation of Organic Sulfides by a Conjugated Polymer as the Photosensitizer for Singlet Oxygen Generation. | Li, J., et al. 2020. ACS Appl Mater Interfaces. 12: 35475-35481. PMID: 32658457
- Synthesis of Two-Dimensional C-C Bonded Truxene-Based Covalent Organic Frameworks by Irreversible Brønsted Acid-Catalyzed Aldol Cyclotrimerization. | Zhang, Q., et al. 2021. Research (Wash D C). 2021: 9790705. PMID: 34549185
- Dual ligand approach increases functional group tolerance in the Pd-catalysed C-H arylation of N-heterocyclic pharmaceuticals. | Beckers, I., et al. 2023. Chem Sci. 14: 1176-1183. PMID: 36756333
- Recent Advances in Chemical Synthesis of Amino Sugars. | Yang, J., et al. 2023. Molecules. 28: PMID: 37375279
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl phenyl sulfide, 5 g | sc-239947 | 5 g | $36.00 |
