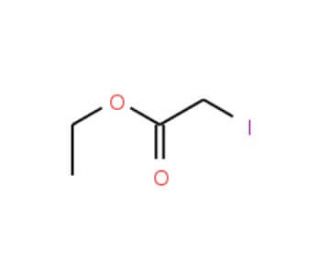
Ethyl iodoacetate (CAS 623-48-3)
QUICK LINKS
Ethyl iodoacetate is a chemical compound that functions as an alkylating agent in development applications. Its mechanism of action involves the alkylation of thiol groups in proteins and enzymes, leading to the inhibition of their activity. Ethyl Iodoacetate is used to modify and study the function of specific proteins and enzymes in experimental applications. Ethyl iodoacetate reacts with thiol groups in a variety of proteins, leading to the disruption of their normal function. Ethyl Iodoacetate is particularly useful for investigating the role of specific thiol-containing proteins in cellular processes. By irreversibly modifying thiol groups, ethyl iodoacetate can provide insights into the molecular mechanisms underlying various biological pathways. Its ability to selectively target thiol-containing proteins may be useful for studying the function of specific enzymes and proteins in experimental. Ethyl iodoacetate′s mechanism of action at the molecular level allows for the precise manipulation and investigation of thiol-containing proteins in various biological systems.
Ethyl iodoacetate (CAS 623-48-3) References
- Effects of parenteral iodoacetate and other thiol reagents on the rabbi retina: relation of histological to biochemical lesions. | NEWHOUSE, JP. and LUCAS, DR. 1959. Br J Ophthalmol. 43: 528-39. PMID: 14426801
- Addition of electrophilic and heterocyclic carbon-centered radicals to glyoxylic oxime ethers. | McNabb, SB., et al. 2004. Org Lett. 6: 1911-4. PMID: 15176781
- Catalytic enantioselective Reformatsky reaction with ketones. | Fernández-Ibáñez, MA., et al. 2008. Chem Commun (Camb). 2571-3. PMID: 18506247
- Catalytic enantioselective Reformatsky reaction with ortho-substituted diarylketones. | Fernández-Ibáñez, MA., et al. 2008. Org Lett. 10: 4041-4. PMID: 18715009
- Mixture toxicity of S(N)2-reactive soft electrophiles: 2-evaluation of mixtures containing ethyl α-halogenated acetates. | Dawson, DA., et al. 2011. Arch Environ Contam Toxicol. 61: 547-57. PMID: 21452006
- Bisoxazolidine-catalyzed enantioselective Reformatsky reaction. | Wolf, C. and Moskowitz, M. 2011. J Org Chem. 76: 6372-6. PMID: 21623640
- Reactions of propargyl compounds containing a cyclobutyl group induced by a ruthenium complex. | Wang, YC., et al. 2012. Chem Asian J. 7: 2703-10. PMID: 22933365
- Synthesis of a leucomitosane via a diastereoselective radical cascade. | Brucelle, F. and Renaud, P. 2013. J Org Chem. 78: 6245-52. PMID: 23721078
- Three-component coupling reactions of arynes for the synthesis of benzofurans and coumarins. | Yoshioka, E., et al. 2014. Molecules. 19: 863-80. PMID: 24419139
- Catalytic Enantioselective Aza-Reformatsky Reaction with Cyclic Imines. | De Munck, L., et al. 2016. Chemistry. 22: 17590-17594. PMID: 27775191
- Novel 6- and 7-Substituted Coumarins with Inhibitory Action against Lipoxygenase and Tumor-Associated Carbonic Anhydrase IX. | Peperidou, A., et al. 2018. Molecules. 23: PMID: 29329232
- Preparation of no carrier added fluorine-18 labeled 16-fluorohexadecanoic and fluoroacetic acids from highly reactive tetraethylammonium [18F]fluoride. | Bosch, AL., et al. 1986. Int J Rad Appl Instrum A. 37: 305-8. PMID: 3021654
- Highly Catalytic Enantioselective Reformatsky Reaction with Aldehydes and Ketones Using an Available Prolinol Ligand. | Ouyang, L., et al. 2020. ACS Omega. 5: 16967-16975. PMID: 32685867
- N-(cyanomethyl)- and N-(2-methoxy-1-cyanoethyl)anthracyclines and related carboxyl derivatives. | Acton, EM., et al. 1986. J Med Chem. 29: 2074-9. PMID: 3761325
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl iodoacetate, 5 g | sc-239931 | 5 g | $32.00 |
