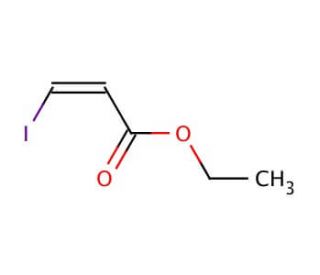
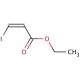
Ethyl cis-3-iodoacrylate (CAS 31930-36-6)
QUICK LINKS
Ethyl cis-3-iodoacrylate also know as (Z)-Ethyl 3-iodoacrylate (EIA), is a colorless liquid with a pungent odor that belongs to the class of acrylates. This organic compound finds extensive use as a reagent in laboratory synthesis, playing a vital role in creating various compounds. EIA holds great significance in the realm of organic synthesis, finding wide applications both in laboratories and industries. The versatility of Ethyl cis-3-iodoacrylate is evident in its scientific research applications. It has contributed to exploring reaction mechanisms and studying the structure and reactivity of organic molecules. The compound has also been instrumental in the synthesis of organic dyes and investigating organic photochemistry. The reactivity of Ethyl cis-3-iodoacrylate arises from its ability to interact with organic molecules. As an alkylating agent, it readily forms covalent bonds by reacting with electron-rich sites on organic molecules. This reaction, known as electrophilic addition, underlies the compound′s reactivity with organic substances.
Ethyl cis-3-iodoacrylate (CAS 31930-36-6) References
- Synthesis of 3,4-disubstituted isoquinolines via palladium-catalyzed cross-coupling of 2-(1-alkynyl)benzaldimines and organic halides. | Dai, G. and Larock, RC. 2003. J Org Chem. 68: 920-8. PMID: 12558417
- Metabolism and elimination of the endogenous DNA adduct, 3-(2-deoxy-beta-D-erythropentofuranosyl)-pyrimido[1,2-alpha]purine-10(3H)-one, in the rat. | Knutson, CG., et al. 2007. J Biol Chem. 282: 36257-64. PMID: 17951255
- Stereo- and regiospecific cu-catalyzed cross-coupling reaction of vinyl iodides and thiols: a very mild and general route for the synthesis of vinyl sulfides. | Kabir, MS., et al. 2008. Org Lett. 10: 3363-6. PMID: 18611035
- A very active cu-catalytic system for the synthesis of aryl, heteroaryl, and vinyl sulfides. | Kabir, MS., et al. 2010. J Org Chem. 75: 3626-43. PMID: 20429581
- Cyclic alkenyl boronic half acid synthesis and applications. | McNulty, L., et al. 2010. J Org Chem. 75: 6001-4. PMID: 20701318
- Palladium membrane-installed microchannel devices for instantaneous Suzuki-Miyaura cross-coupling. | Yamada, YM., et al. 2010. Chemistry. 16: 11311-9. PMID: 20715196
- Selection of monoclonal antibodies against 6-oxo-M(1)dG and their use in an LC-MS/MS assay for the presence of 6-oxo-M(1)dG in vivo. | Akingbade, D., et al. 2012. Chem Res Toxicol. 25: 454-61. PMID: 22211372
- The Synthesis and Bioactivity of the Marine Macrolide Callyspongiolide. | Ko, KY., et al. 2021. Chemistry. 27: 2589-2611. PMID: 32989817
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl cis-3-iodoacrylate, 1 g | sc-235038 | 1 g | $62.00 |
