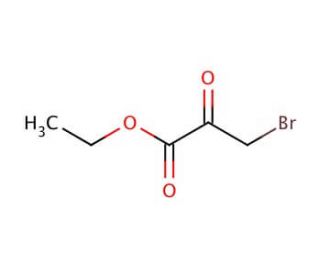
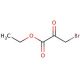
Ethyl bromopyruvate (CAS 70-23-5)
QUICK LINKS
Ethyl bromopyruvate is a brominated derivative of pyruvic acid, featuring an ethyl group esterified to the carboxylic acid, which plays a significant role in synthetic chemistry and biochemical research. This compound serves as a pivotal reagent in the synthesis of a wide array of chemical entities, due to its reactivity particularly in nucleophilic substitution reactions that leverage its halogen atom for the introduction of various functional groups. In research applications, ethyl bromopyruvate is often utilized to study metabolic pathways, where its role in inhibiting certain enzymes involved in glycolysis and the Krebs cycle is of particular interest. This inhibition is valuable in elucidating the mechanisms through which cells manage energy production and regulation, thereby offering insights into the metabolic processes fundamental to cellular function. Its utility extends to the synthesis of heterocyclic compounds, where it acts as a precursor in reactions forming the backbone of many biologically relevant molecules, demonstrating its versatility and importance in both organic synthesis and biochemical investigation.
Ethyl bromopyruvate (CAS 70-23-5) References
- Synthesis of functionalized 5-imino-2,5-dihydro-furans through the reaction of isocyanides with activated acetylenes in the presence of ethyl bromopyruvate. | Yavari, I., et al. 2006. Mol Divers. 10: 479-82. PMID: 16967197
- Variable involvement of the perivascular retinal tissue in carbonic anhydrase inhibitor induced relaxation of porcine retinal arterioles in vitro. | Kehler, AK., et al. 2007. Invest Ophthalmol Vis Sci. 48: 4688-93. PMID: 17898293
- A one-pot catalyst-free synthesis of functionalized pyrrolo[1,2-a]quinoxaline derivatives from benzene-1,2-diamine, acetylenedicarboxylates and ethyl bromopyruvate. | Piltan, M., et al. 2013. Beilstein J Org Chem. 9: 510-5. PMID: 23616791
- The Mycobacterium tuberculosis H37Ra gene MRA_1916 causes growth defects upon down-regulation. | Singh, KS. and Singh, SK. 2015. Sci Rep. 5: 16131. PMID: 26531045
- Sulfuryl transfer catalyzed by pyruvate kinase. | Peliska, JA. and O'Leary, MH. 1989. Biochemistry. 28: 1604-11. PMID: 2719922
- Repurposing ethyl bromopyruvate as a broad-spectrum antibacterial. | Kumar, A., et al. 2019. J Antimicrob Chemother. 74: 912-920. PMID: 30689890
- Synthesis, antioxidant, antimicrobial and antiviral docking studies of ethyl 2-(2-(arylidene)hydrazinyl)thiazole-4-carboxylates. | Haroon, M., et al. 2021. Z Naturforsch C J Biosci. 76: 467-480. PMID: 33901389
- Synthesis, crystal structure, Hirshfeld surface investigation and comparative DFT studies of ethyl 2-[2-(2-nitrobenzylidene)hydrazinyl]thiazole-4-carboxylate. | Haroon, M., et al. 2022. BMC Chem. 16: 18. PMID: 35317817
- Green novel multicomponent synthesis and biological evaluation of new oxazolopyrazoloazepines and reduction of nitrophenols in the presence of Ag/Fe3O4/ZnO@MWCNT MNCs. | Savari, M., et al. 2022. Mol Divers. 26: 3279-3294. PMID: 35322312
- Design, synthesis and evaluation of 2, 6, 8-substituted Imidazopyridine derivatives as potent PI3Kα inhibitors. | Chen, R., et al. 2023. J Enzyme Inhib Med Chem. 38: 2155638. PMID: 36650905
- Research on heterocyclic compounds. XVIII. Imidazo[2,1-b]-1,3,4-thiadiazole derivatives. | Abignente, E., et al. 1985. Farmaco Sci. 40: 190-9. PMID: 3874092
- Synthesis and antitumor activity of 2-beta-D-ribofuranosylselenazole-4- carboxamide and related derivatives. | Srivastava, PC. and Robins, RK. 1983. J Med Chem. 26: 445-8. PMID: 6827564
- Synthesis and pharmacological activity of imidazo[2,1-b]benzothiazole acids. | Grandolini, G., et al. 1993. Farmaco. 48: 31-43. PMID: 8457278
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl bromopyruvate, 5 g | sc-252799 | 5 g | $22.00 | |||
Ethyl bromopyruvate, 25 g | sc-252799A | 25 g | $66.00 |
