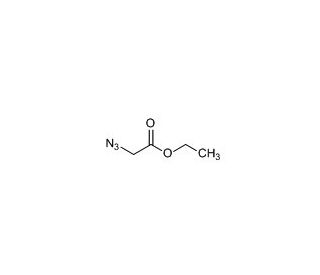
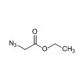
Ethyl Azidoacetate (CAS 637-81-0)
QUICK LINKS
Ethyl azidoacetate is a chemical compound that functions as an alkylating agent in organic synthesis. It acts by undergoing nucleophilic substitution reactions with various nucleophiles, such as amines and thiols, to form new carbon-carbon or carbon-sulfur bonds. Ethyl Azidoacetate′s mechanism of action involves the azido group acting as a leaving group, facilitating the substitution reaction with the nucleophile. Ethyl Azidoacetate′s functional role in development applications involves its use as a reagent for the synthesis of various organic compounds, including intermediates and complex molecules. Its mechanism of action at the molecular level involves the formation of covalent bonds with nucleophilic functional groups, enabling the creation of diverse chemical structures. Ethyl azidoacetate′s role in experimental applications revolves around its ability to facilitate the synthesis of novel organic compounds through its alkylating properties, contributing to the advancement of chemical development.
Ethyl Azidoacetate (CAS 637-81-0) References
- Reusable polymer-supported catalyst for the [3+2] Huisgen cycloaddition in automation protocols. | Girard, C., et al. 2006. Org Lett. 8: 1689-92. PMID: 16597142
- Spatial and temporal control of the alkyne-azide cycloaddition by photoinitiated Cu(II) reduction. | Adzima, BJ., et al. 2011. Nat Chem. 3: 256-59. PMID: 21336334
- A novel copper containing photoinitiator, copper(II) acylphosphinate, and its application in both the photomediated CuAAC reaction and in atom transfer radical polymerization. | Gong, T., et al. 2013. Chem Commun (Camb). 49: 7950-2. PMID: 23900488
- Halide inhibition of the copper-catalysed azide-alkyne cycloaddition. | Moorman, RM., et al. 2015. Org Biomol Chem. 13: 1974-8. PMID: 25537166
- Three-Component Reactions for Post-Polymerization Modifications. | Kakuchi, R. and Theato, P. 2013. ACS Macro Lett. 2: 419-422. PMID: 35581849
- Pyrido annelation reaction by a tandem aza Wittig/electrocyclic ring-closure strategy: Preparation of pyrazolo[4,3-c]- and pyrazolo[3,4-c]pyridine derivatives. | Pedro Molina ∗, Enrique Aller, Angeles Lorenzo. 1991. Tetrahedron. 47: 6737-6746.
- Synthetic studies towards the 2-aminopyrimidine alkaloids variolins and meridianins from marine origin | Pilar M Fresneda, Pedro Molina *, Santiago Delgado, Juan A Bleda. 2000. Tetrahedron Letters. 41: 4777-4780.
- Synthesis of some fused β-carbolines including the first example of the pyrrolo[3,2-c]-β-carboline system | Glenn C. Condie, Jan Bergman. 2004. Journal of Heterocyclic Chemistry. 41: 531-540.
- A simple, efficient thermally promoted protocol for Huisgen-click reaction catalyzed by CuSO4·5H2O in water | Yuqin Jiang a, Duanyang Kong a, Jinglin Zhao a, Weiwei Zhang a, Wenjing Xu b, Wei Li a, Guiqing Xu a. 2014. Tetrahedron Letters. 55: 2410-2414.
- Formation of octaalkylated rctt tetranaphthyl-resorcinarene derivatives containing biologically active components | Valentina V. Glushko, Olga S. Serkova, Vera I. Maslennikova. 2020. Tetrahedron Letters. 61.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl Azidoacetate, 5 g | sc-506144 | 5 g | $95.00 | |||
Ethyl Azidoacetate, 25 g | sc-506144A | 25 g | $275.00 |
