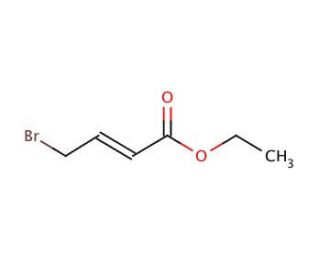
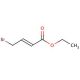
Ethyl 4-bromocrotonate (CAS 37746-78-4)
QUICK LINKS
Ethyl 4-bromocrotonate, referred to as EBC, stands as an organic compound within the ester category. Its chemical structure comprises a crotonic acid group with a bromine atom affixed. Ethyl 4-bromocrotonate′s significance within scientific exploration encompasses a spectrum of aspects. Researchers have delved into its synthesis techniques, mechanism of operation, biochemical and physiological impacts, along with evaluating its pros and cons in laboratory experiments. Moreover, its potential avenues for future exploration have been considered. The applications of ethyl 4-bromocrotonate within scientific investigation extend broadly. It finds utility in scrutinizing biological systems, exploring drug metabolism, and delving into the realm of enzymology. Notably, it functions as a substrate for enzymatic reactions, serving as a reagent for synthesizing diverse organic compounds, and acting as a pivotal tool in the study of enzyme kinetics. A distinctive attribute of ethyl 4-bromocrotonate emerges in its role as an enzyme inhibitor, accomplished through the obstruction of active enzyme sites. By binding to the enzyme′s active site, it effectively thwarts substrate binding, leading to the inhibition of the enzyme′s catalytic function. The imposition of this inhibition can be reversed by introducing a competitive inhibitor, like a substrate analog, thereby restoring the enzyme′s catalytic activity.
Ethyl 4-bromocrotonate (CAS 37746-78-4) References
- New versatile Pd-catalyzed alkylation of indoles via nucleophilic allylic substitution: controlling the regioselectivity. | Bandini, M., et al. 2004. Org Lett. 6: 3199-202. PMID: 15330622
- A short synthesis of (+)-cyclophellitol. | Hansen, FG., et al. 2005. J Org Chem. 70: 10139-42. PMID: 16292857
- Diastereoselective production of homoallylic alcohols bearing quaternary centers from gamma-substituted allylic indiums and ketones. | Babu, SA., et al. 2007. J Org Chem. 72: 10264-7. PMID: 18044919
- Synthesis of casuarine-related derivatives via 1,3-dipolar cycloaddition between a cyclic nitrone and an unsaturated gamma-lactone. | Stecko, S., et al. 2009. Carbohydr Res. 344: 167-76. PMID: 19000623
- Metal nanoparticles or metal oxide nanoparticles, an efficient and promising family of novel heterogeneous catalysts in organic synthesis. | Wang, S., et al. 2009. Dalton Trans. 9363-73. PMID: 19859587
- Development of sensitive direct and indirect enzyme-linked immunosorbent assays (ELISAs) for monitoring bisphenol-A in canned foods and beverages. | Lu, Y., et al. 2012. Anal Bioanal Chem. 403: 1607-18. PMID: 22526648
- Ligand-free Suzuki coupling of arylboronic acids with methyl (E)-4-bromobut-2-enoate: synthesis of unconventional cores of HIV-1 protease inhibitors. | Chiummiento, L., et al. 2012. Org Lett. 14: 3928-31. PMID: 22804107
- Synthesis, receptor binding and activity of iso and azakainoids. | Wang, W., et al. 2013. Bioorg Med Chem Lett. 23: 1949-52. PMID: 23481645
- Asymmetric [3+2] Annulation Approach to 3-Pyrrolines: Concise Total Syntheses of (-)-Supinidine, (-)-Isoretronecanol, and (+)-Elacomine. | Chogii, I. and Njardarson, JT. 2015. Angew Chem Int Ed Engl. 54: 13706-10. PMID: 26447827
- Modular synthesis of thiazoline and thiazole derivatives by using a cascade protocol. | Alsharif, ZA. and Alam, MA. 2017. RSC Adv. 7: 32647-32651. PMID: 29170713
- Thiazolo[3,2-a]pyrimidin-5-one derivatives as a novel class of 11β-hydroxysteroid dehydrogenase inhibitors. | Studzińska, R., et al. 2018. Bioorg Chem. 81: 21-26. PMID: 30086417
- Design and application of intramolecular vinylogous Michael reaction for the construction of 2-alkenyl indoles. | Harish, B., et al. 2021. Chem Commun (Camb). 57: 231-234. PMID: 33300907
- Recent progress toward the asymmetric synthesis of carbon-substituted piperazine pharmacophores and oxidative related heterocycles. | Magriotis, PA. 2020. RSC Med Chem. 11: 745-759. PMID: 33479672
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 4-bromocrotonate, 5 g | sc-239907 | 5 g | $43.00 |
