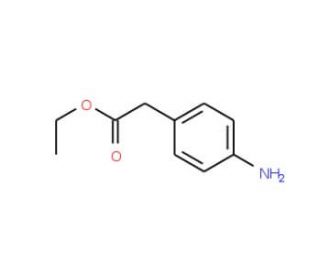
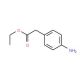
Ethyl 4-aminophenylacetate (CAS 5438-70-0)
QUICK LINKS
Ethyl 4-aminophenylacetate functions as a precursor in organic synthesis. It acts as a building block in the creation of complex molecules, contributing to the synthesis of diverse chemical structures. Ethyl 4-aminophenylacetate′s mechanism of action involves participating in nucleophilic substitution reactions, enabling the introduction of the 4-aminophenylacetate group into target molecules. This allows for the modification of it structure, leading to the formation of new compounds with potential biological activity. Ethyl 4-aminophenylacetate′s role in the experiment involves its incorporation into synthetic pathways, facilitating the production of novel chemical entities for further investigation. Its molecular interactions contribute to the diversification of chemical libraries, supporting the exploration of structure-activity relationships.
Ethyl 4-aminophenylacetate (CAS 5438-70-0) References
- Quinoxaline chemistry. Part 14. 4-(2-Quinoxalylamino)-phenylacetates and 4-(2-quinoxalylamino)-phenylacetyl-L-glutamates as analogues--homologues of classical antifolate agents. Synthesis and evaluation of in vitro anticancer activity. | Piras, S., et al. 2002. Farmaco. 57: 1-8. PMID: 11902640
- Arylacetamide kappa opioid receptor agonists with reduced cytochrome P450 2D6 inhibitory activity. | Le Bourdonnec, B., et al. 2005. Bioorg Med Chem Lett. 15: 2647-52. PMID: 15863335
- Discovery and optimization of ATX inhibitors via modeling, synthesis and biological evaluation. | Balupuri, A., et al. 2018. Eur J Med Chem. 148: 397-409. PMID: 29477073
- Carbocyclic Ring Closure of Aryl C-Glycosides Promoted by Fluoroboric Acid. | Zheng, Q., et al. 2020. J Org Chem. 85: 9339-9346. PMID: 32567318
- Discovery of N-Trisubstituted Pyrimidine Derivatives as Type I RET and RET Gatekeeper Mutant Inhibitors with a Novel Kinase Binding Pose. | Zhang, L., et al. 2022. J Med Chem. 65: 1536-1551. PMID: 35081714
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 4-aminophenylacetate, 5 g | sc-263257 | 5 g | $133.00 |
