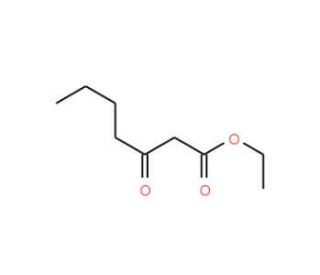
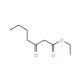
Ethyl 3-Oxoheptanoate (CAS 7737-62-4)
QUICK LINKS
Ethyl 3-Oxoheptanoate, with the CAS number 7737-62-4, is an organic compound categorized as an ester and a ketone, specifically the ethyl ester of 3-oxoheptanoic acid. This molecule features a ketone functional group at the third carbon of a seven-carbon chain, with an ethyl ester at one terminal end. The presence of both a ketone and an ester group in its structure makes Ethyl 3-Oxoheptanoate a valuable reagent in organic synthesis, particularly in the preparation of various intermediates used in the synthesis of more complex chemical structures. The ketone group enhances its reactivity, making it suitable for condensation reactions and nucleophilic additions, which are essential in building cyclic structures and for chain elongation in synthetic pathways. The ester group allows for reactions such as hydrolysis and transesterification, enabling chemists to modify the ester or transform it into other functional groups that are more suited to further synthetic requirements. In research, Ethyl 3-Oxoheptanoate is often employed in the study of Michael addition reactions, where it serves as an electrophilic partner. Additionally, its role in flavor chemistry has been explored, particularly in the synthesis of flavor and fragrance compounds where its degradation or rearrangement under specific conditions can produce desirable flavor profiles. This compound is also used as a reference material in analytical studies for method development and calibration, particularly in systems analyzing ester or ketone functionalities.
Ethyl 3-Oxoheptanoate (CAS 7737-62-4) References
- A new protocol for a regioselective aldol condensation as an alternative convenient synthesis of beta-ketols and alpha,beta-unsaturated ketones. | Kourouli, T., et al. 2002. J Org Chem. 67: 4615-8. PMID: 12076169
- The Pseudomonas aeruginosa autoinducer N-3-oxododecanoyl homoserine lactone accelerates apoptosis in macrophages and neutrophils. | Tateda, K., et al. 2003. Infect Immun. 71: 5785-93. PMID: 14500500
- Tandem oxidation/rearrangement of beta-ketoesters to tartronic esters with molecular oxygen catalyzed by calcium iodide under visible light irradiation with fluorescent lamp. | Kanai, N., et al. 2010. Org Lett. 12: 1948-51. PMID: 20349944
- Design, Synthesis, and Evaluation of Tetrahydropyrrolo[1,2-c]pyrimidines as Capsid Assembly Inhibitors for HBV Treatment. | Li, X., et al. 2017. ACS Med Chem Lett. 8: 969-974. PMID: 28947946
- Synthesis and Pharmacological Evaluation of Triazolopyrimidinone Derivatives as Noncompetitive, Intracellular Antagonists for CC Chemokine Receptors 2 and 5. | Ortiz Zacarías, NV., et al. 2019. J Med Chem. 62: 11035-11053. PMID: 31742400
- The Specificity and Broad Multitarget Properties of Ligands for the Free Fatty Acid Receptors FFA3/GPR41 and FFA2/GPR43 and the Related Hydroxycarboxylic Acid Receptor HCA2/GPR109A. | Bisenieks, E., et al. 2021. Pharmaceuticals (Basel). 14: PMID: 34681211
- Autoregulation of carbapenem biosynthesis in Erwinia carotovora by analogues of N-(3-oxohexanoyl)-L-homoserine lactone. | Chhabra, SR., et al. 1993. J Antibiot (Tokyo). 46: 441-54. PMID: 8478262
- Preparation of Oxepanes, Oxepenes, and Oxocanes by Iodoetherification using Bis(sym-collidine)iodine(I) Hexafluorophosphate as Electrophile | Yves Brunel and Gérard Rousseau. 1996. The Journal of Organic Chemistry. 61: 5793–5800.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 3-Oxoheptanoate, 10 g | sc-294512 | 10 g | $253.00 |
