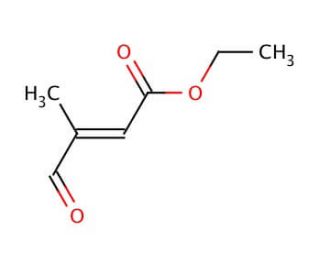
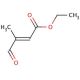
Ethyl 3-methyl-4-oxocrotonate (CAS 62054-49-3)
QUICK LINKS
Ethyl 3-methyl-4-oxocrotonate is a specialized organic compound, a derivative of crotonic acid where the ketone functional group adds significant reactivity. This chemical serves as an important building block in synthetic organic chemistry, particularly in the construction of more complex molecules through various forms of condensation reactions. Its mechanism of action in synthetic applications primarily involves its role as a Michael acceptor due to the presence of the electron-withdrawing ketone group adjacent to a double bond. This configuration makes it highly receptive to nucleophilic attack, facilitating the formation of carbon-carbon bonds in the presence of Michael donors. Research utilizing ethyl 3-methyl-4-oxocrotonate has focused on exploring its utility in facilitating synthetic routes for the creation of diverse organic compounds, including pharmaceuticals and polymers. It is particularly valued for its efficiency in promoting Knoevenagel condensation, a reaction used extensively in the synthesis of various aromatic compounds and heterocycles. Additionally, its use extends to studies in reaction kinetics and mechanisms, where its behavior under different catalytic and environmental conditions can provide insights into the subtleties of reaction pathways and the stability of intermediates formed during synthesis processes. Through these applications, ethyl 3-methyl-4-oxocrotonate contributes significantly to advancements in materials science and medicinal chemistry, where precise manipulation of molecular structures is crucial.
Ethyl 3-methyl-4-oxocrotonate (CAS 62054-49-3) References
- Heteroarotinoids inhibit head and neck cancer cell lines in vitro and in vivo through both RAR and RXR retinoic acid receptors. | Zacheis, D., et al. 1999. J Med Chem. 42: 4434-45. PMID: 10543887
- Aza-retinoids as novel retinoid X receptor-specific agonists. | Farmer, LJ., et al. 2006. Bioorg Med Chem Lett. 16: 2352-6. PMID: 16364638
- Catalytic enantioselective three-component hetero-[4+2] cycloaddition/allylboration approach to alpha-hydroxyalkyl pyrans: scope, limitations, and mechanistic proposal. | Gao, X., et al. 2006. Chemistry. 12: 3132-42. PMID: 16440389
- Organic synthesis of new putative lycopene metabolites and preliminary investigation of their cell-signaling effects. | Reynaud, E., et al. 2011. J Agric Food Chem. 59: 1457-63. PMID: 21247174
- Hyperthermophilic aldolases as biocatalyst for C-C bond formation: rhamnulose 1-phosphate aldolase from Thermotoga maritima. | Oroz-Guinea, I., et al. 2015. Appl Microbiol Biotechnol. 99: 3057-68. PMID: 25324130
- Synthesis of the Bicyclic Lactone Core of Leonuketal, Enabled by a Telescoped Diels-Alder Reaction Sequence. | Grant, PS., et al. 2019. Chem Asian J. 14: 1128-1135. PMID: 30058292
- Development of a Retinal-Based Probe for the Profiling of Retinaldehyde Dehydrogenases in Cancer Cells. | Koenders, STA., et al. 2019. ACS Cent Sci. 5: 1965-1974. PMID: 31893226
- Spectral tuning in bacteriorhodopsin in the absence of counterion and coplanarization effects. | Yan, B., et al. 1995. J Biol Chem. 270: 29668-70. PMID: 8530353
- Synthesis of 2-alkenyl-1-pyrrolin-1-oxides and polysubstituted nitrones. | Sar, Cecilia P., et al. 2003. Synthesis. 2003.09: 1367-1372.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 3-methyl-4-oxocrotonate, 50 ml | sc-257468 | 50 ml | $203.00 |
