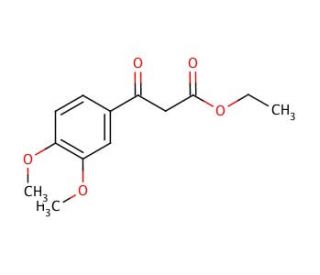
Molecular structure of Ethyl 3,4-dimethoxybenzoylacetate, CAS Number: 4687-37-0
Ethyl 3,4-dimethoxybenzoylacetate (CAS 4687-37-0)
CAS Number:
4687-37-0
Molecular Weight:
252.26
Molecular Formula:
C13H16O5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Ethyl 3,4-dimethoxybenzoylacetate functions as a reagent in organic synthesis. Its mechanism of action involves participating in the Claisen-Schmidt condensation reaction, where it acts as an electrophile, reacting with enolates or enol equivalents to form α,β-unsaturated carbonyl compounds. Ethyl 3,4-Dimethoxybenzoylacetate plays a role in the creation of complex organic molecules by facilitating the formation of new carbon-carbon bonds, which is useful for the development of novel compounds for various research purposes. Its ability to undergo specific chemical reactions may be useful in the exploration of new synthesis pathways and the production of diverse organic compounds.
Ethyl 3,4-dimethoxybenzoylacetate (CAS 4687-37-0) References
- Pyrido[1,2-a]pyrimidin-4-one derivatives as a novel class of selective aldose reductase inhibitors exhibiting antioxidant activity. | La Motta, C., et al. 2007. J Med Chem. 50: 4917-27. PMID: 17845019
- Nanostructured ultra-thin patches for ultrasound-modulated delivery of anti-restenotic drug. | Vannozzi, L., et al. 2016. Int J Nanomedicine. 11: 69-91. PMID: 26730191
- Investigation of HMG-CoA reductase inhibitory and antioxidant effects of various hydroxycoumarin derivatives. | Ozalp, L., et al. 2020. Arch Pharm (Weinheim). 353: e1900378. PMID: 32648617
- Flavonoid Glycosides with a Triazole Moiety for Marine Antifouling Applications: Synthesis and Biological Activity Evaluation. | Pereira, D., et al. 2020. Mar Drugs. 19: PMID: 33374188
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 3,4-dimethoxybenzoylacetate, 1 g | sc-234983 | 1 g | $64.00 |
