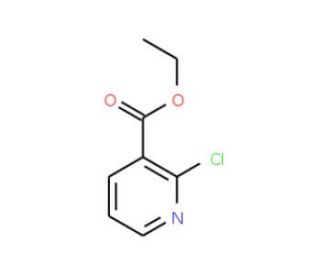
Molecular structure of Ethyl 2-chloronicotinate, CAS Number: 1452-94-4
Ethyl 2-chloronicotinate (CAS 1452-94-4)
Alternate Names:
Ethyl 2-chloropyridine-3-carboxylate; 2-Chloro-3-(ethoxycarbonyl)pyridine
CAS Number:
1452-94-4
Purity:
99%
Molecular Weight:
185.61
Molecular Formula:
C8H8ClNO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Ethyl 2-chloronicotinate functions as a chemical intermediate in organic synthesis. Its mechanism of action involves participating in various chemical reactions, such as esterification, amidation, and nucleophilic substitution, to form new compounds with specific structural and functional properties. At the molecular level, it undergoes transformations to introduce specific functional groups or modify existing ones, enabling the creation of structurally diverse compounds for further investigation and evaluation.
Ethyl 2-chloronicotinate (CAS 1452-94-4) References
- Inhibitors of HCV NS5B polymerase: synthesis and structure-activity relationships of N-1-heteroalkyl-4-hydroxyquinolon-3-yl-benzothiadiazines. | Pratt, JK., et al. 2005. Bioorg Med Chem Lett. 15: 1577-82. PMID: 15745800
- Synthesis and structure-activity relationships of 1,2,4-triazoles as a novel class of potent tubulin polymerization inhibitors. | Ouyang, X., et al. 2005. Bioorg Med Chem Lett. 15: 5154-9. PMID: 16198562
- Toward pyridine-fused selenium-containing antioxidants. | Fenner, T. and Schiesser, CH. 2004. Molecules. 9: 472-9. PMID: 18007447
- High temperature metalation of functionalized aromatics and heteroaromatics using (tmp)2Zn x 2 MgCl2 x 2 LiCl and microwave irradiation. | Wunderlich, S. and Knochel, P. 2008. Org Lett. 10: 4705-7. PMID: 18816136
- Synthesis and anticancer potential of benzothiazole linked phenylpyridopyrimidinones and their diones as mitochondrial apoptotic inducers. | Kamal, A., et al. 2014. Bioorg Med Chem Lett. 24: 147-51. PMID: 24332497
- Design, synthesis and antiproliferative activity of the new conjugates of E7010 and resveratrol as tubulin polymerization inhibitors. | Kamal, A., et al. 2016. Org Biomol Chem. 14: 1382-94. PMID: 26676480
- Mild Cobalt-Catalyzed Negishi Cross-Couplings of (Hetero)arylzinc Reagents with (Hetero)aryl Halides. | Haas, D., et al. 2016. Angew Chem Int Ed Engl. 55: 3809-12. PMID: 26879151
- Preparation of Polyfunctional Organozinc Halides by an InX3 - and LiCl-Catalyzed Zinc Insertion to Aryl and Heteroaryl Iodides and Bromides. | Benischke, AD., et al. 2017. Chemistry. 23: 778-782. PMID: 27862462
- Practical and economic lithiations of functionalized arenes and heteroarenes using Cy2NLi in the presence of Mg, Zn or La halides in a continuous flow. | Becker, MR., et al. 2015. Chem Sci. 6: 6649-6653. PMID: 29435214
- Furopyridines. VI. Preparation and reactions of 2‐and 3‐substituted furo [2, 3‐b] pyridines | Morita, H., & Shiotani, S. 1986. Journal of heterocyclic chemistry. 23(5): 1465-1469.
- Electrosynthesis of functionalized 2-arylpyridines from functionalized aryl and pyridine halides catalyzed by nickel bromide 2, 2′-bipyridine complex | Gosmini, C., Nédélec, J. Y., & Périchon, J. 2000. Tetrahedron Letters. 41(26): 5039-5042.
- Atom‐Economical Preparation of Aryl–and Heteroaryl–Lanthanum Reagents by Directed ortho‐Metalation by Using tmp3 [La] | Wunderlich, S. H., & Knochel, P. 2010. Chemistry–A European Journal. 16(11): 3304-3307.
- Bistrifluoromethylated organocuprate [Ph 4 P]+[Cu (CF 3) 2]−: synthesis, characterization and its application for trifluoromethylation of activated heteroaryl bromides, chlorides and iodides | Liu, H., & Shen, Q. 2019. Organic Chemistry Frontiers. 6(14): 2324-2328.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 2-chloronicotinate, 10 g | sc-263187 | 10 g | $46.00 | |||
Ethyl 2-chloronicotinate, 50 g | sc-263187A | 50 g | $201.00 |
