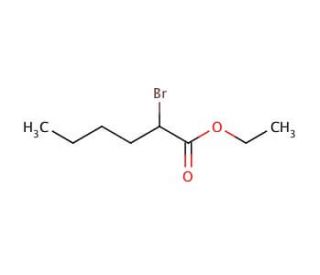
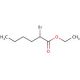
Ethyl 2-bromohexanoate (CAS 615-96-3)
QUICK LINKS
Ethyl 2-bromohexanoate functions as an alkylating agent in chemical reactions, participating in the formation of carbon-carbon bonds. It acts as an electrophile, reacting with nucleophiles to form new carbon-carbon bonds through substitution or addition reactions. Ethyl 2-Bromohexanoate′s mechanism of action involves the displacement of the bromine atom by a nucleophile, leading to the formation of a new carbon-carbon bond. This process is utilized in organic synthesis to introduce the ethyl 2-bromohexanoate moiety into a variety of organic compounds, allowing for the creation of complex molecular structures. In this way, ethyl 2-bromohexanoate plays a role in the construction of diverse chemical entities, contributing to the development of novel compounds for development purposes.
Ethyl 2-bromohexanoate (CAS 615-96-3) References
- Lipophilic methotrexate conjugates with antitumor activity. | Pignatello, R., et al. 2000. Eur J Pharm Sci. 10: 237-45. PMID: 10767601
- Stannyl radical-mediated cleavage of pi-deficient heterocyclic sulfones. Synthesis Of alpha-fluoro esters. | Wnuk, SF., et al. 2000. J Org Chem. 65: 4169-74. PMID: 10866636
- Ni-catalyzed mild arylation of alpha-halocarbonyl compounds with arylboronic acids. | Liu, C., et al. 2007. Org Lett. 9: 5601-4. PMID: 18052180
- Discovery of a novel class of 2-mercaptohexanoic acid derivatives as highly active PPARalpha agonists. | Zettl, H., et al. 2009. Bioorg Med Chem Lett. 19: 4421-6. PMID: 19556125
- Design, synthesis, and biological evaluation of a novel class of gamma-secretase modulators with PPARgamma activity. | Hieke, M., et al. 2010. J Med Chem. 53: 4691-700. PMID: 20503989
- Identification of 2-mercaptohexanoic acids as dual inhibitors of 5-lipoxygenase and microsomal prostaglandin E₂ synthase-1. | Greiner, C., et al. 2011. Bioorg Med Chem. 19: 3394-401. PMID: 21570310
- Studies toward the oxidative and reductive activation of C-S bonds in 2'-S-aryl-2'-thiouridine derivatives. | Rayala, R., et al. 2016. Tetrahedron. 72: 1969-1977. PMID: 27019535
- Nickel-Catalyzed Stereoselective Dicarbofunctionalization of Alkynes. | Li, Z., et al. 2016. Angew Chem Int Ed Engl. 55: 6938-41. PMID: 27111115
- A Haloalkane Dehalogenase from a Marine Microbial Consortium Possessing Exceptionally Broad Substrate Specificity. | Buryska, T., et al. 2018. Appl Environ Microbiol. 84: PMID: 29101190
- Structural and catalytic effects of surface loop-helix transplantation within haloalkane dehalogenase family. | Marek, M., et al. 2020. Comput Struct Biotechnol J. 18: 1352-1362. PMID: 32612758
- Novel thiazolone-benzenesulphonamide inhibitors of human and bacterial carbonic anhydrases. | Abdoli, M., et al. 2023. J Enzyme Inhib Med Chem. 38: 2163243. PMID: 36629426
- Quinoxaline N-oxide containing potent angiotensin II receptor antagonists: synthesis, biological properties, and structure-activity relationships. | Kim, KS., et al. 1993. J Med Chem. 36: 2335-42. PMID: 8360878
- Discovery of OT4003, a novel, potent, and orally active cys-LT1 receptor antagonist. | Tvaermose-Nielsen, O., et al. 1997. Bioorg Med Chem. 5: 415-27. PMID: 9061206
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 2-bromohexanoate, 5 g | sc-234928 | 5 g | $150.00 |
