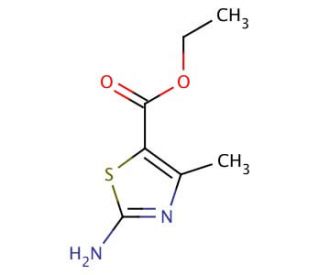
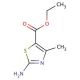
Ethyl-2-amino-4-methyl-thiazole-5-Carboxylate (CAS 7210-76-6)
QUICK LINKS
Ethyl-2-amino-4-methyl-thiazole-5-carboxylate is an organic compound that belongs to the thiazole family, characterized by a five-membered ring containing both sulfur and nitrogen atoms. This compound features an amino group at the 2-position, a methyl group at the 4-position, and an ester group at the 5-position, which contribute to its versatility in chemical reactions and its utility as a synthetic intermediate. The thiazole ring is well-known for its electron-rich nature, making this compound particularly useful in the development of heterocyclic chemistry. In research, ethyl-2-amino-4-methyl-thiazole-5-carboxylate has been employed as a building block for the synthesis of various bioactive molecules and complex heterocycles, especially in the design of novel ligands and catalysts. The amino and ester groups provide reactive sites for further functionalization, enabling the creation of a wide range of derivatives through nucleophilic substitution, amidation, and esterification reactions. Additionally, this compound′s thiazole core is of interest in studying electron delocalization and aromaticity in heterocyclic systems. Researchers have utilized ethyl-2-amino-4-methyl-thiazole-5-carboxylate in the synthesis of materials with potential applications in organic electronics, dyes, and polymers, making it a valuable compound for advancing synthetic methodologies and exploring new chemical frameworks.ι
Ethyl-2-amino-4-methyl-thiazole-5-Carboxylate (CAS 7210-76-6) References
- 2-aminothiazole as a novel kinase inhibitor template. Structure-activity relationship studies toward the discovery of N-(2-chloro-6-methylphenyl)-2-[[6-[4-(2-hydroxyethyl)-1- piperazinyl)]-2-methyl-4-pyrimidinyl]amino)]-1,3-thiazole-5-carboxamide (dasatinib, BMS-354825) as a potent pan-Src kinase inhibitor. | Das, J., et al. 2006. J Med Chem. 49: 6819-32. PMID: 17154512
- Synthesis and biological evaluation of some 2,4,5-trisubstituted thiazole derivatives as potential antimicrobial and anticancer agents. | Al-Saadi, MS., et al. 2008. Arch Pharm (Weinheim). 341: 424-34. PMID: 18574850
- Substituted thiazoles VI. Synthesis and antitumor activity of new 2-acetamido- and 2 or 3-propanamido-thiazole analogs. | El-Messery, SM., et al. 2012. Eur J Med Chem. 54: 615-25. PMID: 22743242
- Synthesis, biological evaluation and molecular modeling study of some new thiazolodiazepine analogs as CNS active agents. | Al-Rashood, STA., et al. 2016. Bioorg Med Chem Lett. 26: 445-453. PMID: 26706170
- Eicosapentaenoic acid metabolism in cynomolgus and rhesus conjunctiva and eyelid. | Kulkarni, PS., et al. 1987. J Ocul Pharmacol. 3: 349-56. PMID: 2846724
- Eicosapentaenoic acid metabolism in human and rabbit anterior uvea. | Kulkarni, PS. and Srinivasan, BD. 1986. Prostaglandins. 31: 1159-64. PMID: 3020617
- Design, synthesis, and bioassay of 4-thiazolinone derivatives as influenza neuraminidase inhibitors. | Xiao, M., et al. 2021. Eur J Med Chem. 213: 113161. PMID: 33540229
- Discovery of 2-(3-Benzamidopropanamido)thiazole-5-carboxylate Inhibitors of the Kinesin HSET (KIFC1) and the Development of Cellular Target Engagement Probes. | Saint-Dizier, F., et al. 2023. J Med Chem. 66: 2622-2645. PMID: 36749938
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl-2-amino-4-methyl-thiazole-5-Carboxylate, 50 µg | sc-205317 | 50 µg | $92.00 | |||
Ethyl-2-amino-4-methyl-thiazole-5-Carboxylate, 100 µg | sc-205317A | 100 µg | $174.00 |
