Ethyl 2,3,4,6-Tetra-O-acetyl-α-D-thiogalactopyranoside (CAS 126187-25-5)
QUICK LINKS
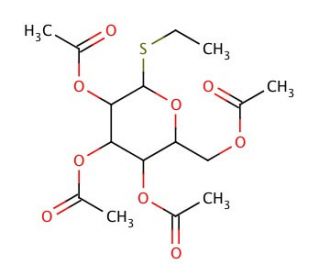
Ethyl 2,3,4,6-Tetra-O-acetyl-α-D-thiogalactopyranoside is a chemically synthesized thioglycoside, which has played a crucial role in carbohydrate chemistry, particularly in glycosylation reactions. This compound features a sulfur atom replacing the oxygen in the glycosidic bond of the sugar, a modification that significantly alters its chemical stability and reactivity, making it resistant to hydrolytic conditions often encountered in organic synthesis. The acetyl groups at the 2, 3, 4, and 6 positions protect the hydroxyl groups of the galactose molecule, facilitating its use in glycosidic bond formation without premature degradation. This protection is essential for the compound′s use in stepwise elongation of carbohydrate chains, where it serves as a glycosyl donor in the presence of promoters such as silver salts or trichloroacetimidate, enabling the formation of glycosidic bonds under controlled conditions. In research, Ethyl 2,3,4,6-Tetra-O-acetyl-α-D-thiogalactopyranoside is particularly valued for its utility in constructing complex oligosaccharides for studies on carbohydrate-protein interactions, which are fundamental to understanding biological recognition processes. These studies contribute significantly to fields like biomaterials science and synthetic biology, providing insights into the synthesis and function of glycomaterials, and enhancing our understanding of biological systems. This compound exemplifies how modifications in sugar chemistry can be leveraged to explore and manipulate biological phenomena in a purely research-focused context.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 2,3,4,6-Tetra-O-acetyl-α-D-thiogalactopyranoside, 2.5 g | sc-221602 | 2.5 g | $380.00 |
