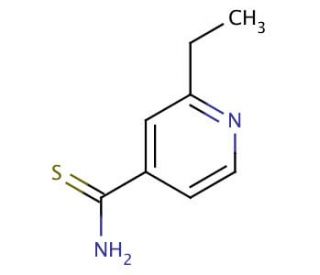
Ethionamide (CAS 536-33-4)
QUICK LINKS
Ethionamide, identified by the CAS number 536-33-4, is a synthetic compound structurally related to thioamide. Its primary mechanism of action lies in its ability to inhibit mycolic acid synthesis, a crucial component of the cell wall of mycobacteria. Ethionamide is activated within bacterial cells by a specific enzyme, EthA, into an active form that targets InhA, an enoyl-ACP reductase involved in fatty acid synthesis. By inhibiting InhA, ethionamide interferes with the production of mycolic acids, thereby disrupting the bacterial cell wall synthesis and compromising the integrity and viability of the bacterial cell. In research settings, ethionamide has been extensively used to study the biochemical pathways of mycobacterial resistance and the pharmacological modulation of antimycobacterial activity. This research has been instrumental in understanding how modifications within the structural design of thioamide derivatives can influence their efficacy against mycobacteria. Additionally, studies involving ethionamide have contributed to the broader field of tuberculosis research by elucidating the genetic mechanisms that mycobacteria employ to develop resistance against antimycobacterial compounds. This has significant implications for the development of new strategies and compounds to combat mycobacterial infections, particularly in strains resistant to conventional treatments.
Ethionamide (CAS 536-33-4) References
- Design, synthesis and molecular docking studies of imidazole and benzimidazole linked ethionamide derivatives as inhibitors of InhA and antituberculosis agents. | Raghu, MS., et al. 2022. Bioorg Med Chem Lett. 60: 128604. PMID: 35123004
- Ethionamide and Prothionamide Based Coumarinyl-Thiazole Derivatives: Synthesis, Antitubercular Activity, Toxicity Investigations and Molecular Docking Studies. | Imran, M. 2022. Pharm Chem J. 56: 1215-1225. PMID: 36531826
- Design, synthesis and anti-Tb evaluation of chalcone derivatives as novel inhibitors of InhA. | L S, D., et al. 2023. J Biomol Struct Dyn. 41: 15165-15176. PMID: 37349907
- Genotypic and phenotypic comparison of drug resistance profiles of clinical multidrug-resistant Mycobacterium tuberculosis isolates using whole genome sequencing in Latvia. | Vīksna, A., et al. 2023. BMC Infect Dis. 23: 638. PMID: 37770850
- Loss-of-function mutations in ndh do not confer delamanid, ethionamide, isoniazid, or pretomanid resistance in Mycobacterium tuberculosis. | Pandey, S., et al. 2024. Antimicrob Agents Chemother. 68: e0109623. PMID: 38038476
- Discovery of dual-active ethionamide boosters inhibiting the Mycobacterium tuberculosis ESX-1 secretion system. | Gries, R., et al. 2024. Cell Chem Biol. 31: 699-711.e6. PMID: 38181799
- Identification of Mycobacterium tuberculosis transcriptional repressor EthR inhibitors: Shape-based search and machine learning studies. | Chikhale, RV., et al. 2024. Heliyon. 10: e26802. PMID: 38434349
- Ethambutol inhibited the growth of acid-fast bacteria and enhanced the detection of Legionella in environmental water samples. | Inoue, H., et al. 2024. J Microorg Control. 29: 1-7. PMID: 38508757
- Crystal structure of the Mycobacterium tuberculosis VirS regulator reveals its interaction with the lead compound SMARt751. | Grosse, C., et al. 2024. J Struct Biol. 216: 108090. PMID: 38548139
- Estimation of country-specific tuberculosis resistance antibiograms using pathogen genomics and machine learning. | Dixit, A., et al. 2024. BMJ Glob Health. 9: PMID: 38548342
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethionamide, 5 g | sc-211429 | 5 g | $250.00 |
