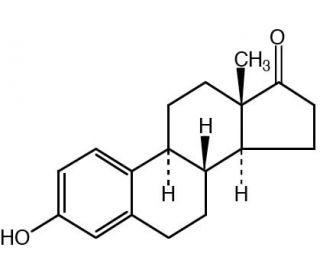
Molecular structure of Estrone, CAS Number: 53-16-7
Estrone (CAS 53-16-7)
Application:
Estrone is an estradiol metabolite and useful in various tumorigenesis studies
CAS Number:
53-16-7
Purity:
>95%
Molecular Weight:
270.37
Molecular Formula:
C18H22O2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Estrone is a estrogen hormone found in both men and women. It is produced in the ovaries in premenopausal women, as well as in the adrenal glands and adipose tissue. In men, it is produced in the testes and adrenal glands. Estrone is one of the three main estrogens produced, along with estradiol and estriol. It plays a role in the regulation of the female reproductive system and is also involved in bone metabolism and cardiovascular health. Estrone is a precursor to estradiol, the most potent estrogen, and can be converted into estradiol. It also has weaker estrogenic activity compared to estradiol. Estrone is an important hormone, contributing to various physiological processes.
Estrone (CAS 53-16-7) References
- Synthesis and antiproliferative activity of epoxy and bromo compounds derived from estrone. | Milic, DR., et al. 2001. Bioorg Med Chem Lett. 11: 2197-200. PMID: 11514169
- Estrone and 17beta-estradiol reverse breast cancer resistance protein-mediated multidrug resistance. | Imai, Y., et al. 2002. Jpn J Cancer Res. 93: 231-5. PMID: 11927002
- Formation of estrone and estradiol from estrone sulfate by normal breast parenchymal tissue. | Chatterton, RT., et al. 2003. J Steroid Biochem Mol Biol. 86: 159-66. PMID: 14568567
- Dietary soy isoflavones and estrone protect ovariectomized ERalphaKO and wild-type mice from carcinogen-induced colon cancer. | Guo, JY., et al. 2004. J Nutr. 134: 179-82. PMID: 14704314
- A total synthesis of estrone based on a novel cascade of radical cyclizations. | Pattenden, G., et al. 2004. Proc Natl Acad Sci U S A. 101: 12024-9. PMID: 15304654
- Influence of dissolved organic matter on estrone removal by NF membranes and the role of their structures. | Jin, X., et al. 2007. Water Res. 41: 3077-88. PMID: 17548103
- Synthesis of 4-formyl estrone using a positional protecting group and its conversion to other C-4-substituted estrogens. | Liu, Y., et al. 2007. J Org Chem. 72: 8824-30. PMID: 17929872
- Evaluation of electrospray ionization and atmospheric pressure chemical ionization for simultaneous detection of estrone and its metabolites using high-performance liquid chromatography/tandem mass spectrometry. | Hsu, JF., et al. 2007. J Chromatogr B Analyt Technol Biomed Life Sci. 860: 49-56. PMID: 17988961
- A molecularly imprinted polymer-coated nanocomposite of magnetic nanoparticles for estrone recognition. | Wang, X., et al. 2009. Talanta. 78: 327-32. PMID: 19203590
- Sorption of estrone and estrone-3-sulfate from CaCl2 solution and artificial urine in pastoral soils of New Zealand. | Scherr, FF., et al. 2009. Environ Toxicol Chem. 28: 2564-71. PMID: 19656001
- Elucidation of the dissociation pathways of electro-ionized estrone. | Bouchonnet, S., et al. 2010. Rapid Commun Mass Spectrom. 24: 973-8. PMID: 20209664
- Photodegradation of estrone enhanced by dissolved organic matter under simulated sunlight. | Caupos, E., et al. 2011. Water Res. 45: 3341-50. PMID: 21530993
- A MALDI-MS-based quantitative analytical method for endogenous estrone in human breast cancer cells. | Kim, KJ., et al. 2016. Sci Rep. 6: 24489. PMID: 27091422
- [Concentrations of testosterone, estrone and estrone sulfate in peripheral blood of donkey stallions in relation to season]. | Schuler, G., et al. 2019. Tierarztl Prax Ausg G Grosstiere Nutztiere. 47: 294-297. PMID: 31634940
- Design and synthesis of hetero-steroids via ring-closing metathesis: Biological studies towards in vitro anticancer activity. | Kotha, S., et al. 2022. Steroids. 188: 109119. PMID: 36202314
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Estrone, 1 g | sc-205683 | 1 g | $33.00 | |||
Estrone, 5 g | sc-205683A | 5 g | $112.00 |
