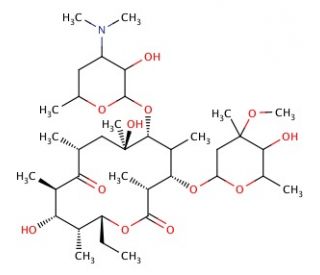
Erythromycin B (CAS 527-75-3)
See product citations (1)
QUICK LINKS
Erythromycin B is a macrocyclic compound and a minor constituent of the erythromycin complex, derived from the fermentation of the bacterium Saccharopolyspora erythraea. Structurally similar to its more prominent counterpart, erythromycin A, erythromycin B features a macrocyclic lactone ring with sugars desosamine and cladinose attached, which are crucial for its bioactivity. The primary mode of action of erythromycin B involves the inhibition of bacterial protein synthesis. It achieves this by binding to the 50S subunit of the bacterial ribosome, thereby blocking the exit tunnel used by the growing peptide chain. This interaction effectively halts the translation process, preventing the assembly of proteins essential for bacterial growth and replication. In research, erythromycin B has been used to study ribosomal structure and function, offering insights into the complex mechanisms of protein synthesis and the role of antibiotics in inhibiting this process. These studies are essential for understanding how ribosomal inhibitors influence bacterial physiology and for the exploration of ribosome-targeting antibiotics. Additionally, erythromycin B serves as a reference molecule in biochemical experiments designed to decipher the nuances of antibiotic action and resistance mechanisms in pathogenic bacteria, thereby contributing to the foundational knowledge in microbial biochemistry.
Erythromycin B (CAS 527-75-3) References
- Acid-catalyzed degradation of clarithromycin and erythromycin B: a comparative study using NMR spectroscopy. | Mordi, MN., et al. 2000. J Med Chem. 43: 467-74. PMID: 10669574
- Microbial degradation of erythromycins A and B. | Flickinger, MC. and Perlman, D. 1975. J Antibiot (Tokyo). 28: 307-11. PMID: 1150530
- Knocking out of tailoring genes eryK and eryG in an industrial erythromycin-producing strain of Saccharopolyspora erythraea leading to overproduction of erythromycin B, C and D at different conversion ratios. | Zhang, Q., et al. 2011. Lett Appl Microbiol. 52: 129-37. PMID: 21175699
- Enhancement of the properties of a drug by mono-deuteriation: reduction of acid-catalysed formation of a gut-motilide enol ether from 8-deuterio-erythromycin B. | Bhadra, PK., et al. 2016. Org Biomol Chem. 14: 6289-96. PMID: 27273525
- Selected Derivatives of Erythromycin B-In Silico and Anti-Malarial Studies. | Bhadra, PK., et al. 2021. Materials (Basel). 14: PMID: 34832380
- Simultaneous determination of erythromycin and its transformation products in treated erythromycin fermentation residue and amended soil. | Feng, H., et al. 2023. Chemosphere. 313: 137414. PMID: 36455662
- Acid degradation of erythromycin A and erythromycin B. | Kurath, P., et al. 1971. Experientia. 27: 362. PMID: 5581079
- Chemistry of erythronolide B. Acid-catalyzed transformations of the aglycone of erythromycin B. | Perun, TJ. 1967. J Org Chem. 32: 2324-30. PMID: 6046179
- Overproduction and characterization of the erythromycin C-12 hydroxylase, EryK. | Lambalot, RH., et al. 1995. Biochemistry. 34: 1858-66. PMID: 7849045
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Erythromycin B, 1 mg | sc-362735 | 1 mg | $98.00 | |||
Erythromycin B, 5 mg | sc-362735A | 5 mg | $490.00 | |||
Erythromycin B, 20 mg | sc-362735B | 20 mg | $1873.00 | |||
Erythromycin B, 50 mg | sc-362735C | 50 mg | $2787.00 | |||
Erythromycin B, 100 mg | sc-362735D | 100 mg | $4815.00 |
