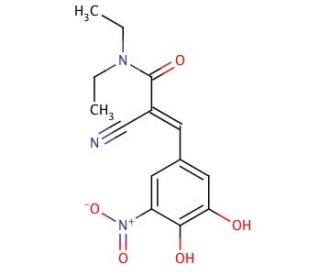
Entacapone (CAS 130929-57-6)
See product citations (1)
QUICK LINKS
Entacapone is a compound extensively utilized in research focused on the inhibition of catechol-O-methyltransferase (COMT), an enzyme involved in the metabolism of catecholamines. In biochemical studies, Entacapone helps to understand the mechanism of COMT and its role in the catabolism of neurotransmitters such as dopamine. It serves as a useful tool in the investigation of the metabolic pathways that influence the duration and effect of certain neurotransmitters in the brain. Additionally, Entacapone is integral to studies examining how the modulation of COMT activity affects the concentration of biologically active catechols in various tissues. In neuroscientific research, it facilitates the exploration of dopaminergic signaling and its implications for motor control and behavior.
Entacapone (CAS 130929-57-6) References
- Entacapone. A review of its use in Parkinson's disease. | Holm, KJ. and Spencer, CM. 1999. Drugs. 58: 159-77. PMID: 10439935
- The role of entacapone in the management of Parkinson's disease. | Brooks, DJ., et al. 2000. Hosp Med. 61: 267-71. PMID: 10858804
- Entacapone. | Chong, BS. and Mersfelder, TL. 2000. Ann Pharmacother. 34: 1056-65. PMID: 10981253
- Entacapone: a catechol-O-methyltransferase inhibitor for the adjunctive treatment of Parkinson's disease. | Najib, J. 2001. Clin Ther. 23: 802-32; discussion 771. PMID: 11440283
- Entacapone/levodopa/carbidopa combination tablet: Stalevo. | . 2003. Drugs R D. 4: 310-1. PMID: 12952501
- Clinical advantages of COMT inhibition with entacapone - a review. | Gordin, A., et al. 2004. J Neural Transm (Vienna). 111: 1343-63. PMID: 15340869
- Entacapone in the treatment of Parkinson's disease. | Schrag, A. 2005. Lancet Neurol. 4: 366-70. PMID: 15907741
- Evidence-based efficacy comparison of tolcapone and entacapone as adjunctive therapy in Parkinson's disease. | Lees, AJ. 2008. CNS Neurosci Ther. 14: 83-93. PMID: 18482101
- Identification of entacapone as a chemical inhibitor of FTO mediating metabolic regulation through FOXO1. | Peng, S., et al. 2019. Sci Transl Med. 11: PMID: 30996080
- Entacapone Treatment Modulates Hippocampal Proteins Related to Synaptic Vehicle Trafficking. | Yoo, DY., et al. 2020. Cells. 9: PMID: 33352833
- Fast Detection of Entacapone by a Lanthanide-Organic Framework with Rhombic Channels. | Min, H., et al. 2021. Chemistry. 27: 17459-17464. PMID: 34608690
- Clinical Pharmacology of Entacapone (Comtan) From the FDA Reviewer. | Habet, S. 2022. Int J Neuropsychopharmacol. 25: 567-575. PMID: 35302623
- Entacapone alleviates acute kidney injury by inhibiting ferroptosis. | Yang, J., et al. 2022. FASEB J. 36: e22399. PMID: 35691001
- [Triple combination of levodopa, carbidopa and entacapone by intrajejunal pump in advanced Parkinson's disease]. | Györfi, B., et al. 2022. Ideggyogy Sz. 75: 365-368. PMID: 36541150
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Entacapone, 10 mg | sc-218315 | 10 mg | $125.00 |
