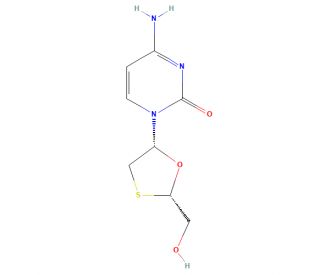
ent-Lamivudine (CAS 134680-32-3)
QUICK LINKS
Ent-Lamivudine, a synthetic nucleoside analogue, is distinct from its parent compound, Lamivudine, due to its chirality. It is primarily utilized in research applications to explore the stereochemical aspects of drug-receptor interactions, highlighting the importance of molecular orientation in biological activity. By mimicking the natural nucleosides that are the building blocks of DNA and RNA, ent-Lamivudine competes with these natural substrates for incorporation into the viral genome. This action is critical for studying the mechanisms by which certain viruses, including retroviruses, replicate their genetic material. When incorporated into the viral DNA during replication, it acts as a chain terminator, preventing the addition of further nucleotides, which halts the replication process. This unique mechanism of action makes ent-Lamivudine useful in the research of viral replication and the development of antiviral strategies. Its role extends beyond merely inhibiting viral replication; it provides insights into the nuances of drug design, particularly in the optimization of chirality for enhancing drug efficacy and specificity.
ent-Lamivudine (CAS 134680-32-3) References
- Chirality Control in Enzyme-Catalyzed Dynamic Kinetic Resolution of 1,3-Oxathiolanes. | Hu, L., et al. 2015. J Org Chem. 80: 8478-81. PMID: 26237578
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
ent-Lamivudine, 1 mg | sc-488694 | 1 mg | $439.00 |
