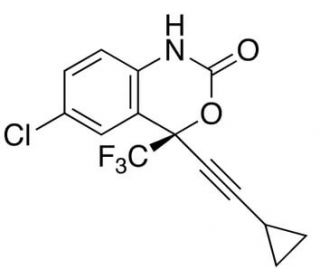
ent Efavirenz (CAS 154801-74-8)
QUICK LINKS
Ent-efavirenz is a stereoisomer of efavirenz, specifically the enantiomer with a reversed chiral configuration compared to the active form commonly studied for its interaction with HIV-1 reverse transcriptase (RT). This difference in stereochemistry leads to distinct properties and interactions with the enzyme. The primary mechanism of action for ent-efavirenz involves the inhibition of HIV-1 reverse transcriptase. Due to its reversed stereochemistry, ent-efavirenz interacts differently with the binding pocket of the RT enzyme, resulting in a different level of inhibitory activity compared to efavirenz. This characteristic makes it useful in comparative studies to understand how stereochemistry affects enzyme binding and activity. In scientific research, ent-efavirenz is utilized to study the structure-function relationship of non-nucleoside inhibitors with the HIV-1 RT enzyme. Researchers use ent-efavirenz to examine how molecular structure modifications can change the binding affinity and efficacy of inhibitors, helping to explain the essential features needed for potent inhibition of the reverse transcriptase enzyme. Furthermore, ent-efavirenz serves as a key compound in exploring resistance mechanisms. By studying how the HIV-1 RT adapts to the presence of an inhibitor with a similar structure but different efficacy, researchers can identify pathways and mutations that contribute to resistance, aiding in the design of more effective inhibitors. Ent-efavirenz is also used in chiral recognition studies. Its distinct stereochemistry allows scientists to investigate chiral discrimination in biological systems, particularly how different enantiomers are metabolized and interact with various enzymes.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
ent Efavirenz, 2.5 mg | sc-207619 | 2.5 mg | $380.00 |
