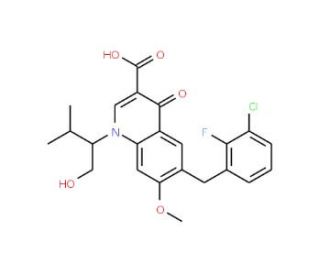
Molecular structure of Elvitegravir, CAS Number: 697761-98-1
Elvitegravir (CAS 697761-98-1)
See product citations (5)
Alternate Names:
6-[(3-chloro-2-fluorophenyl)methyl]-1-[(2S)-1-hydroxy-3-methylbutan-2-yl]-7-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid
Application:
Elvitegravir is An HIV-1 integrase inhibitor.
CAS Number:
697761-98-1
Molecular Weight:
447.88
Molecular Formula:
C23H23ClFNO5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Elvitegravir is an inhibitor of HIV-1 integrase which is responsible for the strand-transfer step of integration. Studies indicate that Elvitegravir is effective against HIV-1 strains that are resistant to other agents such as nucleotide reverse-transcriptase inhibitors (NRTIs), nonnucleoside reverse-transcriptase inhibitors (NNRTIs), and protease inhibitors (PIs), as a result of Elvitegravir′s ability to block the function of HIV1 integrase.
Elvitegravir (CAS 697761-98-1) References
- Elvitegravir: a new HIV integrase inhibitor. | Shimura, K. and Kodama, EN. 2009. Antivir Chem Chemother. 20: 79-85. PMID: 19843978
- Pharmacokinetic interaction of ritonavir-boosted elvitegravir and maraviroc. | Ramanathan, S., et al. 2010. J Acquir Immune Defic Syndr. 53: 209-14. PMID: 19851115
- Activity of elvitegravir, a once-daily integrase inhibitor, against resistant HIV Type 1: results of a phase 2, randomized, controlled, dose-ranging clinical trial. | Zolopa, AR., et al. 2010. J Infect Dis. 201: 814-22. PMID: 20146631
- Elvitegravir overcomes resistance to raltegravir induced by integrase mutation Y143. | Métifiot, M., et al. 2011. AIDS. 25: 1175-8. PMID: 21505303
- In vitro resistance selections using elvitegravir, raltegravir, and two metabolites of elvitegravir M1 and M4. | Margot, NA., et al. 2012. Antiviral Res. 93: 288-296. PMID: 22197635
- Pharmacokinetic profile of raltegravir, elvitegravir and dolutegravir in plasma and mucosal secretions in rhesus macaques. | Massud, I., et al. 2015. J Antimicrob Chemother. 70: 1473-81. PMID: 25630643
- Effects of raltegravir or elvitegravir resistance signature mutations on the barrier to dolutegravir resistance in vitro. | Seki, T., et al. 2015. Antimicrob Agents Chemother. 59: 2596-606. PMID: 25691633
- An unexpected interaction between warfarin and cobicistat-boosted elvitegravir. | Good, BL., et al. 2015. AIDS. 29: 985-6. PMID: 25909832
- HIV integrase inhibitor, Elvitegravir, impairs RAG functions and inhibits V(D)J recombination. | Nishana, M., et al. 2017. Cell Death Dis. 8: e2852. PMID: 28569776
- Degradable bioadhesive nanoparticles for prolonged intravaginal delivery and retention of elvitegravir. | Mohideen, M., et al. 2017. Biomaterials. 144: 144-154. PMID: 28829952
- Spatial distribution of elvitegravir and tenofovir in rat brain tissue: Application of matrix-assisted laser desorption/ionization mass spectrometry imaging and liquid chromatography/tandem mass spectrometry. | Ntshangase, S., et al. 2019. Rapid Commun Mass Spectrom. 33: 1643-1651. PMID: 31240777
- Anti-HIV Drug Elvitegravir Suppresses Cancer Metastasis via Increased Proteasomal Degradation of m6A Methyltransferase METTL3. | Liao, L., et al. 2022. Cancer Res. 82: 2444-2457. PMID: 35507004
- Safety and efficacy of switching to elvitegravir, cobicistat, emtricitabine, tenofovir disoproxil fumarate in treatment-experienced people with HIV: a multicenter cohort study. | De Castro, N., et al. 2023. AIDS Res Ther. 20: 1. PMID: 36597160
Inhibitor of:
HIV-1 Integrase.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Elvitegravir, 10 mg | sc-207615 | 10 mg | $377.00 |
