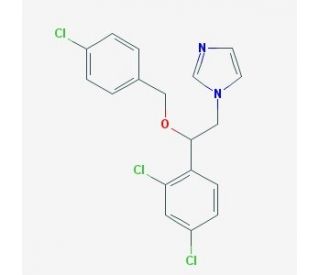
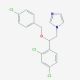
Econazole (CAS 27220-47-9)
QUICK LINKS
Econazole is extensively used in research focusing on antifungal mechanisms and the study of fungal cell biology. In microbiology labs, Econazole is employed to investigate the disruption of fungal membrane integrity and the inhibition of biosynthetic pathways essential for fungal growth. Researchers utilize this compound to study the effects on ergosterol synthesis, a vital component of fungal cell membranes. Additionally, Econazole is used in enzymatic studies to understand its inhibition of fungal enzyme systems, which are for cell wall synthesis and maintenance. This compound also aids in the exploration of resistance mechanisms in pathogenic fungi, providing insights into how these organisms adapt to antifungal agents.
Econazole (CAS 27220-47-9) References
- Econazole induces increases in free intracellular Ca2+ concentrations in human osteosarcoma cells. | Chang, HT., et al. 2005. Hum Exp Toxicol. 24: 453-8. PMID: 16235734
- Econazole attenuates cytotoxicity of 1-methyl-4-phenylpyridinium by suppressing mitochondrial membrane permeability transition. | Lee, CS., et al. 2006. Brain Res Bull. 69: 687-94. PMID: 16716839
- Novel drug delivery strategies for improving econazole antifungal action. | Firooz, A., et al. 2015. Int J Pharm. 495: 599-607. PMID: 26383840
- Econazole imprinted textiles with antifungal activity. | Hossain, MA., et al. 2016. Eur J Pharm Biopharm. 101: 137-44. PMID: 26883854
- Effect of econazole and benzydamine on sensory neurons in culture. | Mathivanan, S., et al. 2016. J Physiol Pharmacol. 67: 851-858. PMID: 28195065
- Econazole nitrate inhibits PI3K activity and promotes apoptosis in lung cancer cells. | Dong, C., et al. 2017. Sci Rep. 7: 17987. PMID: 29269744
- Ophthalmic Econazole Hydrogels for the Treatment of Fungal Keratitis. | Díaz-Tomé, V., et al. 2018. J Pharm Sci. 107: 1342-1351. PMID: 29305870
- Evaluation of topical econazole nitrate formulations with potential for treating Raynaud's phenomenon. | Bahl, D., et al. 2019. Pharm Dev Technol. 24: 689-699. PMID: 30712434
- Stability and toxicity studies for duloxetine and econazole on Spirodela polyrhiza using chiral capillary electrophoresis. | Valimaña-Traverso, J., et al. 2019. J Hazard Mater. 374: 203-210. PMID: 31003121
- Enantioselective determination of econazole in rat plasma and its application to a pharmacokinetic study. | Zhang, J., et al. 2020. Anal Biochem. 602: 113791. PMID: 32473119
- Structural mechanisms of TRPV6 inhibition by ruthenium red and econazole. | Neuberger, A., et al. 2021. Nat Commun. 12: 6284. PMID: 34725357
- Econazole as adjuvant to conventional antibiotics is able to eradicate starvation-induced tolerant bacteria by causing proton motive force dissipation. | Wang, M., et al. 2022. J Antimicrob Chemother. 77: 425-432. PMID: 34747463
- Synergistic Antimicrobial Effect of Colistin in Combination with Econazole against Multidrug-Resistant Acinetobacter baumannii and Its Persisters. | Xie, M., et al. 2022. Microbiol Spectr. 10: e0093722. PMID: 35467374
- Econazole: a review of its antifungal activity and therapeutic efficacy. | Heel, RC., et al. 1978. Drugs. 16: 177-201. PMID: 98315
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Econazole, 5 g | sc-279013 | 5 g | $240.00 |
