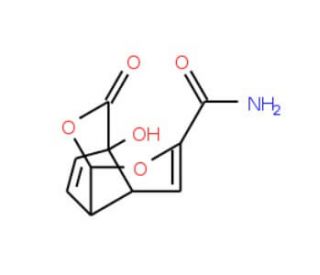
Echinosporin (CAS 79127-35-8)
QUICK LINKS
Echinosporin, identified by the CAS number 79127-35-8, is a chemical compound categorized as an antibiotic and is derived from the actinomycete Actinoplanes echinospora. This compound is notable for its unique structure and biological activity, primarily functioning as an antibacterial agent by targeting and disrupting bacterial cell membranes. The mechanism of action of echinosporin involves the interaction with membrane lipids, leading to the destabilization and increased permeability of bacterial cell membranes. This action results in the leakage of vital cellular contents and ultimately leads to bacterial cell death. In research settings, echinosporin has been utilized to study microbial physiology and membrane dynamics. By examining how echinosporin affects bacterial cell membranes, researchers have gained insights into the essential processes of membrane integrity and the mechanisms by which bacteria resist or succumb to external chemical assaults. Studies involving echinosporin also contribute to a broader understanding of membrane-active compounds and their potential mechanisms of action, providing valuable information for the exploration of novel antibacterial strategies. This compound′s role in disrupting bacterial homeostasis makes it a useful tool for probing the vulnerabilities of bacterial cells and understanding the structural features that contribute to its bioactivity.
Echinosporin (CAS 79127-35-8) References
- Echinosporins as new cell cycle inhibitors and apoptosis inducers from marine-derived Streptomyces albogriseolus. | Cui, CB., et al. 2007. Fitoterapia. 78: 238-40. PMID: 17376609
- Oligomycins and pamamycin homologs impair motility and induce lysis of zoospores of the grapevine downy mildew pathogen, Plasmopara viticola. | Dame, ZT., et al. 2016. FEMS Microbiol Lett. 363: PMID: 27354061
- Echinosporin antibiotics isolated from Amycolatopsis strain and their antifungal activity against root-rot pathogens of the Panax notoginseng. | Xu, X., et al. 2019. Folia Microbiol (Praha). 64: 171-175. PMID: 30117099
- Proteomic analysis reveals the metabolic versatility of Amycolatopsis sp. BX17: A strain native from milpa agroecosystem soil. | Palafox-Félix, M., et al. 2022. J Proteomics. 253: 104461. PMID: 34922014
- Antitumor activity of echinosporin. | Morimoto, M. and Imai, R. 1985. J Antibiot (Tokyo). 38: 490-5. PMID: 4008342
- A new antibiotic echinosporin (XK-213) - producing organism, isolation and characterization. | Sato, T., et al. 1982. J Antibiot (Tokyo). 35: 266-71. PMID: 7076574
- The crystal and molecular structure of echinosporin. | Hirayama and Noriaki, et al. 1983. Bulletin of the Chemical Society of Japan. 56.1: 287-289.
- Biosynthesis of the antibiotic echinosporin by a novel branch of the shikimate pathway. | Dübeler and Anke, et al. 2002. European Journal of Organic Chemistry. 2002.6: 983-987.
- Inhibitory effects of Bacillus licheniformis BL06 on Phytophthora capsici in pepper by multiple modes of action. | Li and Ye, et al. 2020. Biological control. 144: 104210.
- Proteomic and metabolomic analysis reveals that Amycolatopsis sp. BX17 synthesizes antifungal metabolites against Fusarium graminearum through the shikimate pathway. | García-López and Héctor, et al. 2024. Biological Control. 189: 105445.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Echinosporin, 1 mg | sc-358661 | 1 mg | $311.00 | |||
Echinosporin, 5 mg | sc-358661A | 5 mg | $992.00 |
