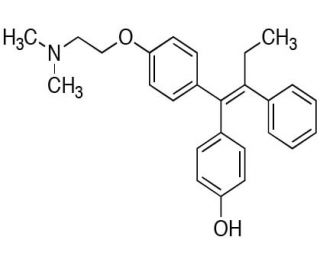
Molecular structure of (E)-4-Hydroxytamoxifen, CAS Number: 174592-47-3
(E)-4-Hydroxytamoxifen (CAS 174592-47-3)
See product citations (1)
Application:
(E)-4-Hydroxytamoxifen is a metabolite of Tamoxifen
CAS Number:
174592-47-3
Purity:
≥97%
Molecular Weight:
387.51
Molecular Formula:
C26H29NO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
(E)-4-Hydroxytamoxifen is an active metabolite of the tamoxifen. This metabolite acts as a selective estrogen receptor modulator, binding to the estrogen receptor and blocking the effects of estrogen in breast tissue. (E)-4-Hydroxytamoxifen is being studied for its potential use in hormone-related conditions. Its ability to modulate estrogen receptor activity makes it important in the study of hormone-related conditions.
(E)-4-Hydroxytamoxifen (CAS 174592-47-3) References
- Synthesis and sulfatase inhibitory activities of (E)- and (Z)-4-hydroxytamoxifen sulfamates. | Chu, GH., et al. 1999. Bioorg Med Chem Lett. 9: 141-4. PMID: 10021916
- Transforming events in thyroid tumorigenesis and their association with follicular lesions. | Heaney, AP., et al. 2001. J Clin Endocrinol Metab. 86: 5025-32. PMID: 11600580
- UDP-glucuronosyltransferase (UGT) 2B15 pharmacogenetics: UGT2B15 D85Y genotype and gender are major determinants of oxazepam glucuronidation by human liver. | Court, MH., et al. 2004. J Pharmacol Exp Ther. 310: 656-65. PMID: 15044558
- Steroid sulfatase: molecular biology, regulation, and inhibition. | Reed, MJ., et al. 2005. Endocr Rev. 26: 171-202. PMID: 15561802
- Synthesis of 1,1,2-triphenylethylenes and their antiproliferative effect on human cancer cell lines. | Zheng, L., et al. 2007. Anticancer Drugs. 18: 1039-44. PMID: 17704654
- Evidence for oxazepam as an in vivo probe of UGT2B15: oxazepam clearance is reduced by UGT2B15 D85Y polymorphism but unaffected by UGT2B17 deletion. | He, X., et al. 2009. Br J Clin Pharmacol. 68: 721-30. PMID: 19916996
- Tamoxifen promotes superoxide production in platelets by activation of PI3-kinase and NADPH oxidase pathways. | Shah, VP., et al. 2012. Thromb Res. 129: 36-42. PMID: 21875743
- Recent advances in the synthesis of raloxifene: a selective estrogen receptor modulator. | Dadiboyena, S. 2012. Eur J Med Chem. 51: 17-34. PMID: 22405286
- Genetic polymorphism of UDP-glucuronosyltransferase (UGT2B15) and glucuronidation of paracetamol in healthy population. | Mehboob, H., et al. 2016. Pak J Pharm Sci. 29: 1037-41. PMID: 27383482
- Nonisomerizable analogues of (Z)- and (E)-4-hydroxytamoxifen. Synthesis and endocrinological properties of substituted diphenylbenzocycloheptenes. | McCague, R., et al. 1988. J Med Chem. 31: 1285-90. PMID: 3385723
- Synthesis, conformational considerations, and estrogen receptor binding of diastereoisomers and enantiomers of 1-[4-[2-(dimethylamino)ethoxy]phenyl]-1,2-diphenylbutane (dihydrotamoxifen). | McCague, R. and Leclercq, G. 1987. J Med Chem. 30: 1761-7. PMID: 3656352
- Structure-activity relationships of selective estrogen receptor modulators: modifications to the 2-arylbenzothiophene core of raloxifene. | Grese, TA., et al. 1997. J Med Chem. 40: 146-67. PMID: 9003514
- Acquired Resistance to Tamoxifen: Back to the Beginning | Maximov, P. Y., McDaniel, R. E., Jordan, V. C., Maximov, P. Y., McDaniel, R. E., & Jordan, V. C. 2013. Tamoxifen: Pioneering Medicine in Breast Cancer,. 143-163.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(E)-4-Hydroxytamoxifen, 5 mg | sc-205662 | 5 mg | $194.00 | |||
(E)-4-Hydroxytamoxifen, 10 mg | sc-205662A | 10 mg | $315.00 |
