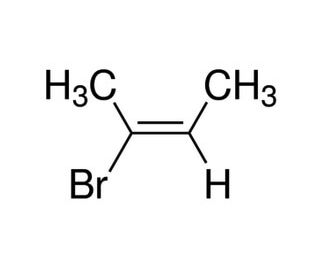
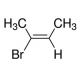
(E)-2-Bromo-2-butene (CAS 3017-71-8)
QUICK LINKS
(E)-2-Bromo-2-butene functions as an alkylating agent. Its mechanism of action involves the ability to form covalent bonds with nucleophilic groups in molecules, particularly with sulfur, oxygen, and nitrogen atoms in proteins and nucleic acids. (E)-2-Bromo-2-Butene can modify the structure and function of biomolecules by alkylating specific sites, leading to changes in their activity and interactions. (E)-2-Bromo-2-butene′s mechanism of action involves the substitution of a bromine atom for a hydrogen atom in the target molecule, resulting in the formation of a new carbon-carbon bond. This process can disrupt the normal function of the biomolecule, providing insights into the role of specific functional groups in biological processes. (E)-2-Bromo-2-butene′s reactivity allows for the selective modification of certain biomolecules.
(E)-2-Bromo-2-butene (CAS 3017-71-8) References
- Hemilabile ligands in organolithium chemistry: substituent effects on lithium ion chelation. | Ramírez, A., et al. 2003. J Am Chem Soc. 125: 15376-87. PMID: 14664582
- Short and efficient total synthesis of fraxinellone limonoids using the stereoselective Oshima-Utimoto reaction. | Trudeau, S. and Morken, JP. 2005. Org Lett. 7: 5465-8. PMID: 16288532
- Concise synthesis of the neo-clerodane skeleton of teucrolivin A using a pericyclic reaction cascade. | Arns, S. and Barriault, L. 2006. J Org Chem. 71: 1809-16. PMID: 16496965
- Synthesis enables a structural revision of the Mycobacterium tuberculosis-produced diterpene, edaxadiene. | Spangler, JE., et al. 2010. Chem Sci. 1: 202-205. PMID: 22114734
- Enantioselective total syntheses and absolute configuration of JBIR-02 and Mer-A2026B. | Hoecker, J. and Gademann, K. 2013. Org Lett. 15: 670-3. PMID: 23330661
- Direct comparison of 3-centre and 4-centre HBr elimination pathways in methyl-substituted vinyl bromides. | Pandit, S., et al. 2016. Phys Chem Chem Phys. 18: 28353-28364. PMID: 27722312
- Synthesis of E- and Z-trisubstituted alkenes by catalytic cross-metathesis. | Nguyen, TT., et al. 2017. Nature. 552: 347-354. PMID: 29293209
- Scalable Synthesis of (-)-Rasfonin Enabled by a Convergent Enantioselective α-Hydroxymethylation Strategy. | Boeckman, RK., et al. 2018. Org Lett. 20: 5062-5065. PMID: 30074398
- Radical Functionalization of Unsaturated Amino Acids: Synthesis of Side-Chain-Fluorinated, Azido-Substituted, and Hydroxylated Amino Acids. | Reeve, PAP., et al. 2019. ACS Omega. 4: 10854-10865. PMID: 31460183
- Catalytic Cross-Metathesis Reactions That Afford E- and Z-Trisubstituted Alkenyl Bromides: Scope, Applications, and Mechanistic Insights. | Koengeter, T., et al. 2023. J Am Chem Soc. 145: 3774-3785. PMID: 36724200
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(E)-2-Bromo-2-butene, 5 ml | sc-234855 | 5 ml | $219.00 |
