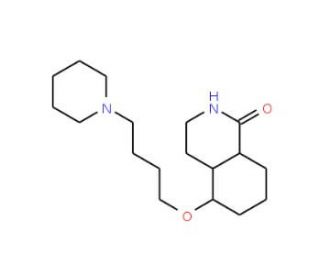
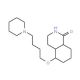
DPQ (CAS 129075-73-6)
See product citations (18)
QUICK LINKS
DPQ is employed in research primarily for its role as a poly(ADP-ribose) polymerase (PARP) inhibitor. This activity is for studies on DNA repair mechanisms, particularly in the context of single-strand DNA breaks where PARP plays a fundamental role. Research involving DPQ often focuses on understanding how inhibition of PARP affects cellular recovery and survival following DNA damage. Additionally, DPQ is used in studies examining the role of PARP in programmed cell death and its implications for cellular stress responses. Furthermore, the compound facilitates investigations into the broader cellular consequences of impaired DNA repair mechanisms, contributing to insights into the stability of the genome and the cellular lifecycle.
DPQ (CAS 129075-73-6) References
- Poly(ADP-ribose) polymerase inhibitors attenuate necrotic but not apoptotic neuronal death in experimental models of cerebral ischemia. | Moroni, F., et al. 2001. Cell Death Differ. 8: 921-32. PMID: 11526447
- Comet assay as a novel approach for studying DNA damage in focal cerebral ischemia: differential effects of NMDA receptor antagonists and poly(ADP-ribose) polymerase inhibitors. | Giovannelli, L., et al. 2002. J Cereb Blood Flow Metab. 22: 697-704. PMID: 12045668
- Clinical perspectives of PARP inhibitors. | Graziani, G. and Szabó, C. 2005. Pharmacol Res. 52: 109-18. PMID: 15911339
- Prolonged poly(ADP-ribose) polymerase-1 activity regulates JP-8-induced sustained cytokine expression in alveolar macrophages. | Espinoza, LA., et al. 2007. Free Radic Biol Med. 42: 1430-40. PMID: 17395016
- Poly(ADP-ribose) polymerase inhibition reduces tumor necrosis factor-induced inflammatory response in rheumatoid synovial fibroblasts. | García, S., et al. 2008. Ann Rheum Dis. 67: 631-7. PMID: 17890271
- Dihydroisoquinolinones: the design and synthesis of a new series of potent inhibitors of poly(ADP-ribose) polymerase. | Suto, MJ., et al. 1991. Anticancer Drug Des. 6: 107-17. PMID: 1903948
- PARP-1 inhibitors DPQ and PJ-34 negatively modulate proinflammatory commitment of human glioblastoma cells. | Scalia, M., et al. 2013. Neurochem Res. 38: 50-8. PMID: 23011206
- PARP-1 inhibitor, DPQ, attenuates LPS-induced acute lung injury through inhibiting NF-κB-mediated inflammatory response. | Wang, G., et al. 2013. PLoS One. 8: e79757. PMID: 24278171
- PARP inhibition attenuates histopathological lesion in ischemia/reperfusion renal mouse model after cold prolonged ischemia. | del Moral, RM., et al. 2013. ScientificWorldJournal. 2013: 486574. PMID: 24319370
- Influence of PARP-1 inhibition in the cardiotoxicity of the topoisomerase 2 inhibitors doxorubicin and mitoxantrone. | Damiani, RM., et al. 2018. Toxicol In Vitro. 52: 203-213. PMID: 29913208
- A novel antagonist of TRPM2 and TRPV4 channels: Carvacrol. | Nazıroğlu, M. 2022. Metab Brain Dis. 37: 711-728. PMID: 34989943
- Poly(ADP-ribose) polymerase gene disruption renders mice resistant to cerebral ischemia. | Eliasson, MJ., et al. 1997. Nat Med. 3: 1089-95. PMID: 9334719
- Role of poly(ADP-ribose) synthetase in inflammation and ischaemia-reperfusion. | Szabó, C. and Dawson, VL. 1998. Trends Pharmacol Sci. 19: 287-98. PMID: 9703762
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
DPQ, 1 mg | sc-202755 | 1 mg | $66.00 | |||
DPQ, 5 mg | sc-202755A | 5 mg | $256.00 |
