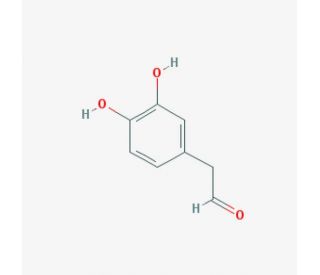
Molecular structure of Dopal, CAS Number: 5707-55-1
Dopal (CAS 5707-55-1)
See product citations (14)
Alternate Names:
3,4-dihydroxyphenylacetaldehyde
Application:
Dopal is a metabolite of the neurotransmitter dopamine
CAS Number:
5707-55-1
Molecular Weight:
152.15
Molecular Formula:
C8H8O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
DOPAL is an aldehyde product from the oxidative deamination of dopamine by monoamine oxidase. Dopal, an organic compound present in various organisms like bacteria, fungi, and plants, falls under the category of aldehydes and serves as a precursor to the neurotransmitter dopamine. Its significance lies in its application within research focused on understanding the molecular intricacies of neurological disorders like Parkinson′s disease, schizophrenia, and depression. Furthermore, Dopal has proven valuable in investigating the impact of oxidative stress on cellular function, as well as unraveling the role of dopamine in processes such as learning and memory.
Dopal (CAS 5707-55-1) References
- Vesicular uptake blockade generates the toxic dopamine metabolite 3,4-dihydroxyphenylacetaldehyde in PC12 cells: relevance to the pathogenesis of Parkinson's disease. | Goldstein, DS., et al. 2012. J Neurochem. 123: 932-43. PMID: 22906103
- Aldehyde dehydrogenase inhibition as a pathogenic mechanism in Parkinson disease. | Fitzmaurice, AG., et al. 2013. Proc Natl Acad Sci U S A. 110: 636-41. PMID: 23267077
- Divalent metal ions enhance DOPAL-induced oligomerization of alpha-synuclein. | Jinsmaa, Y., et al. 2014. Neurosci Lett. 569: 27-32. PMID: 24670480
- Benomyl, aldehyde dehydrogenase, DOPAL, and the catecholaldehyde hypothesis for the pathogenesis of Parkinson's disease. | Casida, JE., et al. 2014. Chem Res Toxicol. 27: 1359-61. PMID: 25045800
- Oligomerization and Membrane-binding Properties of Covalent Adducts Formed by the Interaction of α-Synuclein with the Toxic Dopamine Metabolite 3,4-Dihydroxyphenylacetaldehyde (DOPAL). | Follmer, C., et al. 2015. J Biol Chem. 290: 27660-79. PMID: 26381411
- Toxic Dopamine Metabolite DOPAL Forms an Unexpected Dicatechol Pyrrole Adduct with Lysines of α-Synuclein. | Werner-Allen, JW., et al. 2016. Angew Chem Int Ed Engl. 55: 7374-8. PMID: 27158766
- Antioxidant-Mediated Modulation of Protein Reactivity for 3,4-Dihydroxyphenylacetaldehyde, a Toxic Dopamine Metabolite. | Anderson, DG., et al. 2016. Chem Res Toxicol. 29: 1098-107. PMID: 27268734
- DOPAL derived alpha-synuclein oligomers impair synaptic vesicles physiological function. | Plotegher, N., et al. 2017. Sci Rep. 7: 40699. PMID: 28084443
- Isoindole Linkages Provide a Pathway for DOPAL-Mediated Cross-Linking of α-Synuclein. | Werner-Allen, JW., et al. 2018. Biochemistry. 57: 1462-1474. PMID: 29394048
- Exploring the role of methionine residues on the oligomerization and neurotoxic properties of DOPAL-modified α-synuclein. | Carmo-Gonçalves, P., et al. 2018. Biochem Biophys Res Commun. 505: 295-301. PMID: 30249394
- Aldehyde adducts inhibit 3,4-dihydroxyphenylacetaldehyde-induced α-synuclein aggregation and toxicity: Implication for Parkinson neuroprotective therapy. | Kumar, VB., et al. 2019. Eur J Pharmacol. 845: 65-73. PMID: 30579934
- Formation of large oligomers of DOPAL-modified α-synuclein is modulated by the oxidation of methionine residues located at C-terminal domain. | Coelho-Cerqueira, E., et al. 2019. Biochem Biophys Res Commun. 509: 367-372. PMID: 30591215
- Enhanced mitochondrial inhibition by 3,4-dihydroxyphenyl-acetaldehyde (DOPAL)-oligomerized α-synuclein. | Sarafian, TA., et al. 2019. J Neurosci Res. 97: 1689-1705. PMID: 31420910
- In Vitro Protective Action of Monomeric and Fibrillar α-Synuclein on Neuronal Cells Exposed to the Dopaminergic Toxins Salsolinol and DOPAL. | Carmo-Gonçalves, P., et al. 2020. ACS Chem Neurosci. 11: 3541-3548. PMID: 33080132
- Parkinson-like early autonomic dysfunction induced by vagal application of DOPAL in rats. | Sun, J., et al. 2021. CNS Neurosci Ther. 27: 540-551. PMID: 33475253
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dopal, 25 mg | sc-391117 | 25 mg | $1248.00 | |||
Dopal, 250 mg | sc-391117A | 250 mg | $4682.00 | |||
Dopal, 1 g | sc-391117B | 1 g | $8843.00 |
