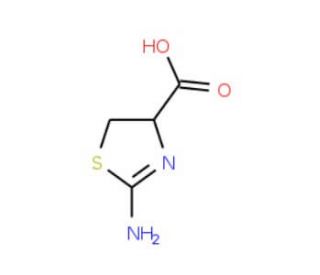
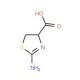
DL-2-Amino-2-thiazoline-4-carboxylic acid (CAS 2150-55-2)
QUICK LINKS
DL-2-Amino-2-thiazoline-4-carboxylic acid finds widespread utility in various scientific experiments and research endeavors. It plays a role in biochemical and physiological investigations, as well as in the exploration of enzymes and proteins. Additionally, it contributes significantly to the study of cell signaling pathways. The primary function of DL-2-Amino-2-thiazoline-4-carboxylic acid lies in its inhibitory properties towards enzymes and proteins. By selectively binding to the active site of these biomolecules, it effectively impedes their normal functionality. Moreover, this compound also interacts with the cell membrane, obstructing the reception of external signals by the cells. Through these inhibitory actions, DL-2-Amino-2-thiazoline-4-carboxylic acid plays a vital role in elucidating molecular mechanisms and physiological processes.
DL-2-Amino-2-thiazoline-4-carboxylic acid (CAS 2150-55-2) References
- Identification, cloning, and sequencing of the genes involved in the conversion of D,L-2-amino-delta2-thiazoline-4-carboxylic acid to L-cysteine in Pseudomonas sp. strain ON-4a. | Ohmachi, T., et al. 2002. Biosci Biotechnol Biochem. 66: 1097-104. PMID: 12092821
- Biotechnological production of amino acids and derivatives: current status and prospects. | Leuchtenberger, W., et al. 2005. Appl Microbiol Biotechnol. 69: 1-8. PMID: 16195792
- Metabolic control analysis of L-cysteine producing strain TS1138 of Pseudomonas sp. | Huai, L., et al. 2009. Biochemistry (Mosc). 74: 288-92. PMID: 19364323
- Graphene-like nets of hydrogen-bonded water molecules in the dihydrate of 2-[(2-ammonioethyl)amino]acetate and the structure of its anhydrous hydroiodide salt. | Wiklund, T., et al. 2010. Acta Crystallogr C. 66: o410-3. PMID: 20679718
- Isolation and genetic improvement of Pseudomonas sp. strain HUT-78, capable of enzymatic production of L-cysteine from DL-2-amino-Δ2-thiazoline-4-carboxylic acid. | Yang, B., et al. 2011. J Gen Appl Microbiol. 57: 379-86. PMID: 22353743
- Enhanced biocatalytic production of L-cysteine by Pseudomonas sp. B-3 with in situ product removal using ion-exchange resin. | Wang, P., et al. 2015. Bioprocess Biosyst Eng. 38: 421-8. PMID: 25199811
- Enzymes for pharmaceutical and therapeutic applications. | Meghwanshi, GK., et al. 2020. Biotechnol Appl Biochem. 67: 586-601. PMID: 32248597
- Fungal lipases as biocatalysts: A promising platform in several industrial biotechnology applications. | Mahfoudhi, A., et al. 2022. Biotechnol Bioeng. 119: 3370-3392. PMID: 36137755
- Metabolic stress constrains microbial L-cysteine production in Escherichia coli by accelerating transposition through mobile genetic elements. | Heieck, K., et al. 2023. Microb Cell Fact. 22: 10. PMID: 36642733
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
DL-2-Amino-2-thiazoline-4-carboxylic acid, 1 g | sc-285456 | 1 g | $221.00 | |||
DL-2-Amino-2-thiazoline-4-carboxylic acid, 5 g | sc-285456A | 5 g | $856.00 |
