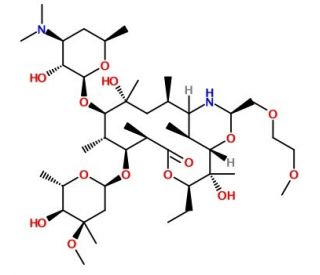
Dirithromycin (CAS 62013-04-1)
QUICK LINKS
Dirithromycin, cataloged under CAS number 62013-04-1, is a macrolide antibiotic derivative known for its unique prodrug form, which upon administration is metabolized into its active component, erythromycylamine. It has been a focal point in non-clinical microbiological research due to its novel mode of action and transformation within biological systems. The primary mechanism of dirithromycin involves the inhibition of protein synthesis in bacterial cells. It achieves this by binding to the bacterial 50S ribosomal subunit, a critical component of the protein synthesis machinery. This binding impedes the translocation step of protein elongation, which is essential for the growth and replication of bacteria. This specific interaction disrupts the production of essential proteins, leading to bacterial growth inhibition and eventual bacterial cell death. In research settings, dirithromycin has been extensively used to study the dynamics of macrolide action, particularly its binding affinity and specificity towards the ribosomal subunit. Studies have also focused on the metabolic transformation of dirithromycin into its active form, providing insights into prodrug activation mechanisms that could influence the design of future antibiotics. Additionally, research involving dirithromycin has contributed to understanding how bacteria develop resistance to macrolides, which is critical for advancing knowledge on combating antibiotic resistance in various bacterial strains.
Dirithromycin (CAS 62013-04-1) References
- In vitro effects of spiramycin and dirithromycin on IL1 beta production by human LPS-stimulated mononuclear cells. | Moutard, I., et al. 1999. Therapie. 54: 607-12. PMID: 10667098
- Efficacy of oral levofloxacin and dirithromycin alone and in combination with rifampicin in the treatment of experimental murine Brucella abortus infection. | Arda, B., et al. 2004. Int J Antimicrob Agents. 23: 204-7. PMID: 15013048
- Characterization of impurities in dirithromycin by liquid chromatography/ion trap mass spectrometry. | Diana, J., et al. 2006. J Chromatogr A. 1125: 52-66. PMID: 16782117
- Development and validation of an improved liquid chromatographic method for the analysis of dirithromycin. | Diana, J., et al. 2006. Talanta. 70: 1064-72. PMID: 18970882
- Determination of aminoglycoside and macrolide antibiotics in meat by pressurized liquid extraction and LC-ESI-MS. | Berrada, H., et al. 2010. J Sep Sci. 33: 522-9. PMID: 20155746
- Insights into the improved macrolide inhibitory activity from the high-resolution cryo-EM structure of dirithromycin bound to the E. coli 70S ribosome. | Pichkur, EB., et al. 2020. RNA. 26: 715-723. PMID: 32144191
- Development of a latex particles-based lateral flow immunoassay for group determination of macrolide antibiotics in breast milk. | Raysyan, A., et al. 2020. J Pharm Biomed Anal. 189: 113450. PMID: 32693204
- Immunotechniques for the Group Determination of Macrolide Antibiotics Traces in the Environment Using a Volume-Mediated Sensitivity Enhancement Strategy. | Burkin, MA., et al. 2023. Biosensors (Basel). 13: PMID: 37887114
- Potentiometric planar platforms modified with a multiwalled carbon nanotube/polyaniline nanocomposite and based on imprinted polymers for erythromycin assessment. | Almehizia, AA., et al. 2023. RSC Adv. 13: 35926-35936. PMID: 38090101
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Dirithromycin, 1 g | sc-211359 | 1 g | $205.00 |
