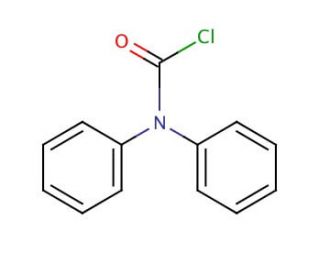
Diphenylcarbamoyl chloride (CAS 83-01-2)
QUICK LINKS
Diphenylcarbamoyl chloride is a chemical compound that functions as an acylating agent in organic synthesis. It is used to introduce the diphenylcarbamoyl group into various organic molecules, allowing for the modification of their chemical properties. The mode of action of diphenylcarbamoyl chloride involves the reaction of the carbonyl chloride group with nucleophilic functional groups, such as amines or alcohols, to form amides or esters, respectively. This acylation reaction occurs through the displacement of the chloride ion, resulting in the formation of a covalent bond between the diphenylcarbamoyl group and the nucleophilic substrate. Diphenylcarbamoyl chloride modification can be utilized to alter the reactivity, solubility, or other properties of the target molecules for further experimental manipulation. In this way, diphenylcarbamoyl chloride serves as a function to synthesize and modification of organic compounds.
Diphenylcarbamoyl chloride (CAS 83-01-2) References
- Studies on the synthesis of a G-rich octaoligoisonucleotide (isoT)2(isoG)4(isoT)2 by the phosphotriester approach and its formation of G-quartet structure. | Chen, J., et al. 2002. Nucleic Acids Res. 30: 3005-14. PMID: 12087187
- Carbonic anhydrase inhibitors. Inhibition of cytosolic isozymes I and II and transmembrane, cancer-associated isozyme IX with lipophilic sulfonamides. | Franchi, M., et al. 2003. J Enzyme Inhib Med Chem. 18: 333-8. PMID: 14567548
- The base pairing properties of 8-aza-7-deaza-2'-deoxyisoguanosine and 7-halogenated derivatives in oligonucleotide duplexes with parallel and antiparallel chain orientation. | Seela, F. and Kröschel, R. 2003. Nucleic Acids Res. 31: 7150-8. PMID: 14654690
- A new, but old, nucleoside analog: the first synthesis of 1-deaza-2'-deoxyguanosine and its properties as a nucleoside and as oligodeoxynucleotides. | Kojima, N., et al. 2003. Nucleic Acids Res. 31: 7175-88. PMID: 14654693
- Skeletal keratan sulphate chains isolated from bovine intervertebral disc may terminate in alpha(2----6)-linked N-acetylneuraminic acid. | Dickenson, JM., et al. 1992. Biochem J. 282 (Pt 1): 267-71. PMID: 1540142
- Synthesis and characterization of modified nucleotides in the 970 hairpin loop of Escherichia coli 16S ribosomal RNA. | Abeydeera, ND. and Chow, CS. 2009. Bioorg Med Chem. 17: 5887-93. PMID: 19628400
- Two linkage-region fragments isolated from skeletal keratan sulphate contain a sulphated N-acetylglucosamine residue. | Dickenson, JM., et al. 1990. Biochem J. 269: 55-9. PMID: 2142881
- p21-Activated kinase 2 (PAK2) inhibits TGF-β signaling in Madin-Darby canine kidney (MDCK) epithelial cells by interfering with the receptor-Smad interaction. | Yan, X., et al. 2012. J Biol Chem. 287: 13705-12. PMID: 22393057
- The medicinal chemistry of imidazotetrazine prodrugs. | Moody, CL. and Wheelhouse, RT. 2014. Pharmaceuticals (Basel). 7: 797-838. PMID: 25014631
- Isoguanine and 5-methyl-isocytosine bases, in vitro and in vivo. | Bande, O., et al. 2015. Chemistry. 21: 5009-22. PMID: 25684598
- Mechanistic Studies of the Solvolyses of Carbamoyl Chlorides and Related Reactions. | D'Souza, MJ. and Kevill, DN. 2016. Int J Mol Sci. 17: PMID: 26784185
- Synthesis of Oligonucleotides Containing the N6 -(2-Deoxy-α,β-d-erythropentofuranosyl)-2,6-diamino-4-hydroxy-5-formamidopyrimidine (Fapy⋅dG) Oxidative Damage Product Derived from 2'-Deoxyguanosine. | Yang, H., et al. 2020. Chemistry. 26: 5441-5448. PMID: 32271495
- Overcoming GNA/RNA base-pairing limitations using isonucleotides improves the pharmacodynamic activity of ESC+ GalNAc-siRNAs. | Schlegel, MK., et al. 2021. Nucleic Acids Res. 49: 10851-10867. PMID: 34648028
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Diphenylcarbamoyl chloride, 25 g | sc-239803 | 25 g | $50.00 |
